Armodafinil
The R-enantiomer of modafinil, a eugeroic (wakefulness-promoting agent) prescribed for narcolepsy, shift work sleep disorder, and obstructive sleep apnea-related sleepiness.
Overview
Armodafinil is the purified (R)-enantiomer of modafinil, belonging to the benzhydryl class of compounds and categorized as a eugeroic — a class of wakefulness-promoting agents pharmacologically distinct from traditional psychostimulants like amphetamines. Marketed under the brand name Nuvigil by Cephalon (now Teva Pharmaceutical), it received FDA approval in 2007 for the treatment of excessive daytime sleepiness associated with narcolepsy, obstructive sleep apnea, and shift work sleep disorder. Compared to racemic modafinil, armodafinil achieves higher plasma concentrations later in the day due to its longer elimination half-life of approximately 15 hours.
The precise mechanism of action remains incompletely understood, but the primary pharmacological target appears to be the dopamine transporter (DAT). Armodafinil acts as a selective, atypical dopamine reuptake inhibitor, increasing extracellular dopamine concentrations particularly in prefrontal cortical regions. This dopaminergic activity triggers downstream effects including activation of wake-promoting orexinergic neurons in the hypothalamus and inhibition of GABAergic transmission in the ventrolateral preoptic nucleus, a key sleep-promoting region. Additional modulation of noradrenergic and histaminergic systems also contributes to its wakefulness-promoting profile.
Armodafinil is also used off-label for attention deficit hyperactivity disorder, chronic fatigue syndrome, major depressive disorder, and as adjunctive therapy in bipolar depression. While it shares the stimulant class with amphetamines from a regulatory standpoint (Schedule IV controlled substance in the United States), it has a substantially lower abuse potential and does not produce the pronounced euphoria associated with classical psychostimulants. Common side effects include headache, nausea, dizziness, and insomnia. It is primarily metabolized via amide hydrolysis and CYP3A4/5-mediated sulfone formation, and may interact with hormonal contraceptives and other CYP3A4 substrates.
Mechanism of Action
Dopamine Transporter Inhibition
Armodafinil is the (R)-enantiomer of modafinil, a eugeroic (wakefulness-promoting) agent. Its primary mechanism involves selective inhibition of the dopamine transporter (DAT), increasing extracellular dopamine concentrations in the nucleus accumbens, prefrontal cortex, and striatum. Armodafinil binds to DAT at an atypical site distinct from classical psychostimulants like amphetamine, producing a slower onset and lower abuse potential (PMID: 19037200).
Orexin/Hypocretin System Activation
Armodafinil activates orexinergic neurons in the lateral hypothalamus, increasing orexin-A and orexin-B signaling through OX1R and OX2R receptors. This stimulates the tuberomammillary nucleus (histaminergic), locus coeruleus (noradrenergic), and dorsal raphe (serotonergic) arousal centers. The orexin-dependent component is critical — armodafinil shows diminished efficacy in orexin-deficient narcolepsy models (PMID: 22050227).
Noradrenergic & Histaminergic Pathways
Downstream of DAT inhibition and orexin activation, armodafinil increases norepinephrine release in the cortex via alpha-1 adrenergic receptor activation and elevates histamine levels in the tuberomammillary nucleus. Both pathways converge on cortical arousal circuits, sustaining wakefulness through enhanced thalamocortical signaling (PMID: 18784649).
GABAergic Suppression
Armodafinil indirectly reduces GABA release in the ventrolateral preoptic area (VLPO), the key sleep-promoting nucleus. By diminishing GABAergic inhibition of wake-active centers, armodafinil shifts the sleep-wake balance toward sustained arousal without the rebound hypersomnia characteristic of amphetamines (PMID: 16076930).
Pharmacokinetic Advantage
The (R)-enantiomer has a longer effective half-life (~15 h vs ~4 h for the S-enantiomer), producing more sustained plasma concentrations and maintaining wakefulness through late afternoon without evening dosing.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Potent at Lower Doses:: Typically effective at 150mg vs 200mg modafinil.. Genetic Variation:: Effectiveness may vary based on CYP450 enzyme genetics.
- Typically effective at 150mg vs 200mg modafinil.
- Effectiveness may vary based on CYP450 enzyme genetics.
Safety Profile
Common Side Effects
- Headache (most common, reported in ~34% of patients per FDA label)
- Nausea and dizziness
- Insomnia and difficulty falling asleep
- Dry mouth
- Anxiety and nervousness
- Diarrhea and upper abdominal pain
- Palpitations
Serious Adverse Reactions (per FDA label)
- Serious skin reactions: Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) reported with modafinil/armodafinil. Discontinue immediately at first sign of rash.
- Multi-organ hypersensitivity (DRESS syndrome): Potentially fatal; presents with fever, rash, lymphadenopathy, and organ involvement
- Angioedema and anaphylaxis: Cases reported post-marketing
- Psychiatric symptoms: Mania, delusions, hallucinations, suicidal ideation; monitor closely in patients with psychiatric history
- Cardiovascular: Not recommended in patients with mitral valve prolapse or left ventricular hypertrophy. Cases of chest pain and palpitations reported.
Contraindications (FDA label - NUVIGIL)
- Known hypersensitivity to armodafinil or modafinil
- History of serious dermatologic reactions to either agent
Drug Interactions
- CYP3A4/5 substrates (cyclosporine, midazolam, triazolam): Armodafinil is a moderate CYP3A4 inducer; may reduce efficacy
- CYP2C19 inhibition: May increase levels of omeprazole, phenytoin, diazepam
- Hormonal contraceptives: Reduced efficacy; alternative or additional contraception recommended during and for one month after discontinuation (FDA label)
- Warfarin: Monitor INR; potential for altered anticoagulant response
- MAOIs: Use with caution
Special Populations
- Pregnancy: FDA former Category C. Insufficient human data; animal studies showed developmental toxicity. A pregnancy registry existed (closed).
- Pediatric: Not approved for pediatric use. Serious skin reactions reported in pediatric patients receiving modafinil.
- Geriatric: Consider lower doses; hepatic clearance may be reduced.
- Hepatic impairment: Reduce dose in severe hepatic impairment (FDA label).
- Schedule IV controlled substance: Risk of abuse and dependence.
Monitoring
- Blood pressure and heart rate at baseline and periodically
- Skin assessments, especially in first weeks of therapy
- Psychiatric symptom monitoring
- Hepatic function in patients with liver disease
- Pregnancy testing in women of childbearing potential
Pharmacokinetic Profile
Armodafinil — Pharmacokinetic Curve
SubcutaneousQuick Start
- Typical Dose
- 150mg once daily in the morning.
Molecular Structure
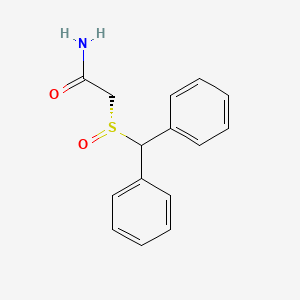
- Formula
- C15H15NO2S
- Weight
- 273.4 Da
- PubChem CID
- 9690109
- Exact Mass
- 273.0823 Da
- LogP
- 1.7
- TPSA
- 79.4 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 3
- Rotatable Bonds
- 5
- Complexity
- 302
Identifiers (SMILES, InChI)
InChI=1S/C15H15NO2S/c16-14(17)11-19(18)15(12-7-3-1-4-8-12)13-9-5-2-6-10-13/h1-10,15H,11H2,(H2,16,17)/t19-/m1/s1
YFGHCGITMMYXAQ-LJQANCHMSA-NSafety Profile
Common Side Effects
- Headaches:: Common, often mitigated with hydration.
- Appetite Suppression:: Significant reduction in hunger.
- Insomnia:: Can disrupt sleep if taken too late.
- Dry Mouth:: Increased thirst common.
References (7)
- [1]Armodafinil as a Potential Pharmacological Treatment for Attention Deficit Hyperactivity Disorder in Adults: A Review
→ Armodafinil demonstrates potential as a treatment for adult ADHD, improving wakefulness, memory, impulse control, and executive functions.
- [2]Adjunctive armodafinil for major depressive episodes associated with bipolar I disorder
→ At 150 mg/day, adjunctive armodafinil improved depressive symptoms in bipolar depression patients, though not all measures showed improvement.
- [3]Adjunct armodafinil improves wakefulness and memory in obstructive sleep apnea/hypopnea syndrome
→ Armodafinil enhanced wakefulness, memory, and reduced fatigue in obstructive sleep apnea patients while maintaining regular nCPAP use.
- [4]Armodafinil as adjunctive therapy in adults with cognitive deficits associated with schizophrenia
→ At 200 mg/day, armodafinil showed no cognitive improvement but appeared to mitigate negative schizophrenia symptoms.
- [5]Armodafinil for treatment of excessive sleepiness associated with shift work disorder
→ Armodafinil significantly improved wakefulness during night shifts and overall clinical condition without affecting daytime sleep.
- [6]Efficacy of Treatment With Armodafinil for Cancer-Related Fatigue in Patients With High-grade Glioma
→ Armodafinil showed no significant reduction in cancer-related fatigue in high-grade glioma patients, but may benefit corticosteroid users.
- [7]A phase 3, double-blind, randomized, placebo-controlled study of armodafinil for excessive sleepiness associated with jet lag disorder
→ At 150 mg/day, armodafinil significantly increased wakefulness and reduced sleep latency following eastward travel across 6 time zones.
Argireline (Acetyl Hexapeptide-8)
Argireline (acetyl hexapeptide-8) is a synthetic peptide derived from the N-terminal end of SNAP-25 protein that works as a topical alternative to botulinum tox
Artemisinin
Artemisinin is a sesquiterpene lactone compound extracted from Artemisia annua (sweet wormwood), originally developed as an antimalarial drug. It works through