Sodium Ascorbate
A buffered, non-acidic mineral salt form of vitamin C that provides the same antioxidant and immune-supporting benefits as ascorbic acid with significantly improved gastrointestinal tolerance and neutral pH.
Overview
Sodium ascorbate is the sodium salt of ascorbic acid (vitamin C), formed by reacting ascorbic acid with sodium bicarbonate or sodium hydroxide. The resulting compound has a near-neutral pH of approximately 7.4, compared to the strongly acidic pH of 2.4 for ascorbic acid, making it substantially gentler on the gastrointestinal tract. This buffered form provides identical ascorbate bioavailability and antioxidant activity as free ascorbic acid once absorbed, while virtually eliminating the gastric irritation, heartburn, and diarrhea that limit high-dose ascorbic acid supplementation in many individuals. Each gram of sodium ascorbate delivers approximately 889 mg of ascorbic acid equivalent and 111 mg of sodium.
As a source of vitamin C, sodium ascorbate participates in all the same biochemical functions: it serves as a cofactor for prolyl and lysyl hydroxylases in collagen synthesis, supports carnitine biosynthesis required for mitochondrial fatty acid transport, enhances non-heme iron absorption, regenerates oxidized vitamin E back to its active form, and supports immune function through enhancement of neutrophil chemotaxis, phagocytosis, and lymphocyte proliferation. The ascorbate moiety also acts as a potent water-soluble antioxidant, scavenging superoxide, hydroxyl radicals, and hypochlorous acid while protecting lipid membranes from peroxidative damage in concert with glutathione and the thioredoxin system.
Sodium ascorbate is widely used in high-dose vitamin C protocols where gastrointestinal tolerance is a limiting factor, including intravenous vitamin C (IVC) preparations used in integrative oncology and sepsis research. For oral supplementation, it is preferred by individuals who experience stomach upset with ascorbic acid, particularly at doses exceeding 1-2 grams daily. The sodium content is minimal at standard supplemental doses but should be considered in sodium-restricted diets at very high doses. Other buffered vitamin C forms include calcium ascorbate, magnesium ascorbate, and the lipid-soluble ascorbyl palmitate, each offering different advantages depending on the clinical context and patient needs.
Mechanism of Action
Mechanism of Action
Sodium ascorbate (C6H7NaO6) is the sodium salt of L-ascorbic acid with a pH of 7.0-7.5 in solution, compared to pH 2.4 for ascorbic acid. It dissociates in aqueous solution to release sodium ions and ascorbate anions, which are the biologically active form of vitamin C at physiological pH. Since ascorbic acid is >99.9% ionized to ascorbate at pH 7.4, sodium ascorbate and ascorbic acid have identical biological activity once absorbed.
Collagen Biosynthesis
Ascorbate is an absolute requirement for collagen synthesis, serving as a cofactor for prolyl-4-hydroxylase (P4H) and lysyl hydroxylase (PLOD). These Fe2+/2-oxoglutarate-dependent dioxygenases hydroxylate proline (to 4-hydroxyproline) and lysine residues in nascent procollagen chains. 4-Hydroxyproline forms interchain hydrogen bonds that stabilize the collagen triple helix; without hydroxylation, collagen denatures at body temperature. Ascorbate maintains the catalytic iron in the reduced Fe2+ state after each catalytic cycle. This mechanism underlies collagen types I-IV synthesis in skin, bone, blood vessels, cartilage, and basement membranes.
Antioxidant Network
Ascorbate is the primary water-soluble antioxidant in human plasma and intracellular fluid. Its redox potential (E0' = +0.06V for ascorbate/monodehydroascorbate couple) allows it to reduce virtually all biologically relevant radicals while being non-reactive enough to avoid indiscriminate reactions. The monodehydroascorbate radical produced is exceptionally stable and is recycled back to ascorbate by monodehydroascorbate reductase (NADH-dependent) and thioredoxin reductase. Ascorbate also regenerates alpha-tocopherol at the water-membrane interface, linking water-soluble and lipid-soluble antioxidant defenses.
Immune Function
Ascorbate accumulates in immune cells (particularly neutrophils, monocytes, and lymphocytes) via sodium-dependent vitamin C transporter 2 (SVCT2). During infection, neutrophils consume massive amounts of ascorbate during the oxidative burst, using it to protect their own membranes from HOCl and superoxide while maintaining microbicidal activity. Ascorbate also enhances T-cell differentiation by supporting TET enzyme activity that demethylates genes controlling T-helper cell lineage commitment. NK cell cytotoxicity and antibody production are also ascorbate-dependent.
Epigenetic Demethylation
A more recently discovered function of ascorbate is as a cofactor for TET1/2/3 dioxygenases and Jumonji-domain histone demethylases. TET enzymes catalyze the sequential oxidation of 5-methylcytosine (5mC) to 5-hydroxymethylcytosine (5hmC), 5-formylcytosine, and 5-carboxylcytosine, leading to passive and active DNA demethylation. This epigenetic function is critical for embryonic stem cell pluripotency, hematopoietic stem cell self-renewal, and reactivation of silenced tumor suppressor genes. Ascorbate deficiency leads to DNA hypermethylation patterns associated with aging and cancer.
Buffered Formulation Advantage
Sodium ascorbate provides 111 mg of ascorbate per 131 mg (the sodium adds ~11 mg sodium per 1000 mg dose). The neutral pH avoids the gastric irritation, acid reflux, and diarrhea that can occur with high-dose ascorbic acid. Absorption is identical to ascorbic acid via intestinal SVCT1 transporters, with bioavailability of 70-90% at doses up to 200 mg, declining to ~50% at 1000 mg and ~25% at 6000 mg due to transporter saturation.
Research
Reported Effects
Athletic Performance:: Research consistently shows minimal to no benefit for athletic performance, with some studies suggesting high-dose supplementation may actually impair training adaptations. Immune Function:: Effectiveness for preventing illness is modest at best, with most benefit seen in reducing symptom duration by 10-20% rather than prevention. Dosage Dependency:: The RDA of 60-90mg is sufficient for 90% of healthy people, with benefits above this level being unclear and potentially counterproductive. Form Matters:: Sodium ascorbate is preferred by some users for being less acidic and gentler on the stomach compared to regular ascorbic acid
- Research consistently shows minimal to no benefit for athletic performance, with some studies suggesting high-dose supplementation may actually impair training adaptations
- Effectiveness for preventing illness is modest at best, with most benefit seen in reducing symptom duration by 10-20% rather than prevention
- The RDA of 60-90mg is sufficient for 90% of healthy people, with benefits above this level being unclear and potentially counterproductive
- Sodium ascorbate is preferred by some users for being less acidic and gentler on the stomach compared to regular ascorbic acid
Safety Profile
Safety Profile: Sodium Ascorbate
Common Side Effects
- Gastrointestinal symptoms: nausea, abdominal cramps, diarrhea, and bloating (less GI irritation than ascorbic acid due to buffered pH, but still dose-dependent; typically above 2 g/day)
- Mild heartburn (less than with ascorbic acid but possible at high doses)
- Increased urination (mild diuretic effect)
- False-positive glucose urine tests (interferes with some dipstick methods)
Serious Adverse Effects
- Kidney stones: vitamin C is metabolized to oxalate; chronic high-dose intake (>1 g/day) increases urinary oxalate excretion and calcium oxalate kidney stone risk; the NHANES-linked Swedish study showed a 2-fold increased stone risk in men taking >1 g/day vitamin C
- Sodium load: each gram of sodium ascorbate contains ~111 mg sodium; high-dose supplementation (multiple grams/day) contributes significant dietary sodium; relevant for hypertension, heart failure, and sodium-restricted diets
- Iron overload: vitamin C dramatically enhances non-heme iron absorption and can mobilize stored iron; in hemochromatosis or iron overload conditions, this can accelerate organ damage
- Hemolytic anemia in G6PD deficiency: high-dose vitamin C (especially IV) can trigger oxidative hemolysis in individuals with glucose-6-phosphate dehydrogenase deficiency
- Rebound scurvy: abrupt cessation after chronic high-dose supplementation may cause temporary vitamin C deficiency symptoms due to upregulated metabolic clearance
- Interference with lab tests: can cause falsely low blood glucose readings on certain glucometer models and interfere with fecal occult blood testing
Contraindications
- History of calcium oxalate kidney stones (use only at low doses with urologist guidance)
- Hemochromatosis or iron overload disorders
- G6PD deficiency (avoid high doses, especially IV)
- Severe renal insufficiency (impaired oxalate clearance increases stone and oxalosis risk)
- Sodium-restricted diets if using high doses (consider ascorbic acid or calcium ascorbate instead)
Drug Interactions
- Iron supplements: vitamin C increases non-heme iron absorption 2–6 fold; beneficial in iron deficiency but dangerous in iron overload
- Warfarin: high-dose vitamin C (>1 g/day) may reduce warfarin efficacy; monitor INR
- Estrogen-containing contraceptives: vitamin C may increase estrogen levels by inhibiting sulfation; theoretical increase in thrombotic risk at high doses
- Aspirin and NSAIDs: vitamin C and aspirin compete for renal tubular reabsorption; aspirin may decrease vitamin C levels; vitamin C may increase aspirin levels
- Chemotherapy: antioxidant vitamin C may interfere with oxidative mechanisms of certain chemotherapeutics (alkylating agents, anthracyclines); always consult oncologist
- Aluminum-containing antacids: vitamin C increases aluminum absorption; avoid combination in renal insufficiency
Population-Specific Considerations
- General supplementation: RDA is 75–90 mg/day; sodium ascorbate is preferred over ascorbic acid for individuals with sensitive stomachs; typical supplemental doses of 250–500 mg/day are safe for most adults
- Pregnancy: RDA increases to 85 mg/day; supplementation at standard doses is safe; avoid megadoses (>2 g/day) which may cause neonatal rebound scurvy
- Elderly: generally well-tolerated; kidney stone risk and sodium load are primary concerns; maintain hydration
- Hypertension / heart failure: sodium content becomes clinically relevant at doses >2 g/day; consider ascorbic acid or calcium ascorbate formulations instead
- Cancer patients: high-dose IV vitamin C is an area of active research; oral sodium ascorbate is not a substitute; do not use as cancer treatment without oncologist supervision
Pharmacokinetic Profile
Quick Start
- Typical Dose
- 500-1000mg daily is the most commonly reported dose among users, though this exceeds the RDA significantly
Molecular Structure
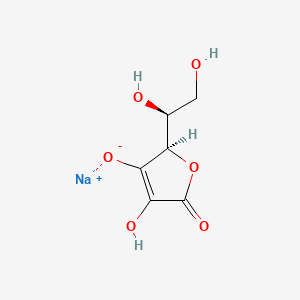
- Formula
- C6H7NaO6
- Weight
- 198.11 Da
- PubChem CID
- 23667548
- Exact Mass
- 198.0140 Da
- TPSA
- 110 Ų
- H-Bond Donors
- 3
- H-Bond Acceptors
- 6
- Rotatable Bonds
- 2
- Complexity
- 237
Identifiers (SMILES, InChI)
InChI=1S/C6H8O6.Na/c7-1-2(8)5-3(9)4(10)6(11)12-5;/h2,5,7-10H,1H2;/q;+1/p-1/t2-,5+;/m0./s1
PPASLZSBLFJQEF-RXSVEWSESA-MSafety Profile
Common Side Effects
- Kidney Stones:: Multiple users report developing kidney stones from taking 1+ grams daily, described as extremely painful and leading to discontinuation
- Digestive Issues:: Some users experience stomach upset or diarrhea, particularly with ascorbic acid forms, leading them to switch to sodium ascorbate or buffered versions
- Training Interference:: Research indicates high-dose antioxidant supplementation may blunt beneficial adaptations to exercise by neutralizing signaling molecules
- Minimal Issues at Moderate Doses:: Most users taking 500-1000mg report no significant side effects when using sodium ascorbate or buffered forms
References (6)
- [5]Enhanced Vitamin C Delivery: A Systematic Literature Review Assessing the Efficacy and Safety of Alternative Supplement Forms in Healthy Adults
→ Systematic review evaluating alternative vitamin C formulations (liposomal, calcium ascorbate, slow-release) compared to traditional ascorbic acid, assessing bioavailability, retention, and effects on immunity in healthy adults.
- [4]Vitamin C and E supplementation hampers cellular adaptation to endurance training in humans
→ Double-blind controlled trial found that vitamin C and E supplementation during endurance training may interfere with beneficial cellular adaptations to exercise in healthy adults.
- [2]Antioxidants and Exercise Performance: With a Focus on Vitamin E and C Supplementation
→ Antioxidant supplementation including vitamin C may impair training adaptations by blocking beneficial signaling from free radicals. Evidence suggests supplementation does not benefit most athletes except those training at altitude or seeking immediate short-term performance enhancement.
- [3]Micronutrients and athletic performance: A review
→ Review of micronutrient supplementation in athletes shows vitamin C is commonly used to correct deficiencies and enhance recovery, though optimal dosing and effectiveness for performance enhancement remains unclear.
- [6]Vitamin C-Sources, Physiological Role, Kinetics, Deficiency, Use, Toxicity, and Determination
→ Comprehensive review of vitamin C covering its essential role as an antioxidant, involvement in immune function and infection resistance, and the importance of supplementation when dietary intake is insufficient.
- [1]Vitamin C Supplementation and Athletic Performance: A Review
→ High-dose vitamin C supplementation shows mixed results for athletic performance, with only 3 of 14 studies showing positive outcomes. Most studies found neutral or negative effects on muscle damage, physical performance, and training adaptations, leading to recommendations against long-term high-dose supplementation.
SNAP-8
SNAP-8, also known as acetyl octapeptide-3, is a synthetic anti-wrinkle peptide composed of eight amino acids. It is an elongated analogue of acetyl hexapeptide
Sodium Bicarbonate
Sodium bicarbonate (baking soda) is an alkaline compound that buffers acid accumulation in muscles during high-intensity exercise by increasing blood bicarbonat