Colchicine
Colchicine is an alkaloid derived from the autumn crocus (Colchicum autumnale) that has been used for centuries to treat gout and is now recognized for its broader anti-inflammatory properties. It works by inhibiting microtubule polymerization and suppressing neutrophil-driven inflammatory cascades.
Overview
Colchicine is a tricyclic alkaloid originally extracted from Colchicum autumnale (autumn crocus or meadow saffron), with a documented therapeutic history spanning over 3,000 years. It was first described in the Ebers Papyrus for joint swelling and was used in ancient Greek and Roman medicine. Today, colchicine is an FDA-approved pharmaceutical agent primarily prescribed for acute gout flares, gout prophylaxis, and familial Mediterranean fever (FMF).
The primary mechanism of action involves binding to tubulin and inhibiting microtubule polymerization, which disrupts multiple cellular processes including neutrophil chemotaxis, adhesion, and recruitment to sites of inflammation. By suppressing neutrophil activation and reducing the release of inflammatory mediators, colchicine effectively dampens the intense inflammatory response characteristic of gout attacks driven by monosodium urate crystal deposition. It also inhibits the NLRP3 inflammasome, reducing interleukin-1β production, a key cytokine in the inflammatory cascade.
Recent cardiovascular research has generated significant interest in colchicine as a cardioprotective agent. The landmark COLCOT and LoDoCo2 trials demonstrated that low-dose colchicine (0.5 mg daily) significantly reduced the risk of cardiovascular events in patients with coronary artery disease, leading to FDA approval for atherosclerotic cardiovascular disease prevention. This application leverages the role of inflammation in atherosclerosis. Colchicine has a narrow therapeutic index, and side effects—primarily gastrointestinal—are dose-dependent. It requires careful dosing and monitoring, particularly in patients with renal or hepatic impairment, and has significant drug interactions with CYP3A4 inhibitors and P-glycoprotein inhibitors.
Mechanism of Action
Tubulin-Binding Alkaloid — Microtubule Depolymerization
Colchicine is a tricyclic tropolone alkaloid isolated from Colchicum autumnale (autumn crocus) that binds with high affinity (Kd ~10 nM) to the colchicine-binding site at the intradimer interface between alpha- and beta-tubulin heterodimers. This binding traps tubulin in a curved conformation incompatible with microtubule lattice incorporation, preventing the addition of new tubulin subunits to growing microtubule plus-ends. At therapeutic concentrations (0.5-2 mg/day), colchicine does not fully depolymerize existing microtubules but rather suppresses dynamic instability — the stochastic switching between growth and shrinkage phases — effectively freezing microtubule dynamics and disrupting microtubule-dependent cellular processes (PMID: 22289869).
Neutrophil Migration & NLRP3 Inflammasome Inhibition
Colchicine's anti-inflammatory mechanism centers on disrupting neutrophil chemotaxis and adhesion. By inhibiting microtubule dynamics, colchicine prevents polarization of neutrophil cytoskeleton, impairs integrin clustering (LFA-1, Mac-1) required for endothelial adhesion, blocks L-selectin expression on the neutrophil surface, and inhibits superoxide production by NADPH oxidase assembly. Critically, colchicine inhibits NLRP3 inflammasome activation by disrupting microtubule-mediated transport of mitochondria to the endoplasmic reticulum, preventing ASC oligomerization and subsequent caspase-1 activation and IL-1beta/IL-18 maturation (PMID: 24790427).
Gout & Crystal Arthropathy Mechanism
In gout, monosodium urate (MSU) crystals are phagocytosed by macrophages, activating the NLRP3 inflammasome through lysosomal rupture and potassium efflux. Colchicine blocks this cascade at multiple points: preventing MSU crystal phagocytosis (microtubule-dependent), inhibiting NLRP3 assembly, and reducing neutrophil recruitment through E-selectin downregulation on endothelial cells (PMID: 25205464).
Cardiovascular Anti-Inflammatory Effects
The COLCOT and LoDoCo2 trials demonstrated colchicine (0.5 mg/day) reduces major cardiovascular events by 23-31% in patients with coronary artery disease, mediated through suppression of residual inflammatory risk — reduction of CRP, IL-6, and IL-1beta that drive atherosclerotic plaque instability (PMID: 31733140).
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Acute Gout:: Highly effective when taken at the very first sign of a flare-up. Cardiovascular Protection:: Shows promise in reducing risk for patients with high cardiovascular inflammation. Therapeutic Window:: Effectiveness is limited by a very narrow range between a healing dose and a toxic dose
- Highly effective when taken at the very first sign of a flare-up
- Shows promise in reducing risk for patients with high cardiovascular inflammation
- Effectiveness is limited by a very narrow range between a healing dose and a toxic dose
Safety Profile
Safety Profile: Colchicine (FDA Approved)
Overview Colchicine is an FDA-approved anti-inflammatory medication primarily used for gout and familial Mediterranean fever. It has a narrow therapeutic index, meaning the difference between therapeutic and toxic doses is small. Careful dosing and monitoring are essential. Brand names include Colcrys and Mitigare.
Common Side Effects
- Gastrointestinal effects are the most frequent: diarrhea (up to 23% of patients), nausea, vomiting, and abdominal pain
- These GI effects are often the first sign of toxicity and serve as a warning to reduce dose
- Loss of appetite and abdominal cramping
- Fatigue and muscle weakness at higher doses
Serious Adverse Effects
- Bone marrow suppression: pancytopenia, agranulocytosis, thrombocytopenia, and aplastic anemia, which can be fatal
- Myopathy and rhabdomyolysis: particularly when combined with statins or in renal impairment
- Peripheral neuropathy: may develop with chronic use
- Multi-organ failure: reported in overdose situations, often fatal at doses exceeding 0.5 mg/kg
- Disseminated intravascular coagulation (DIC): in severe toxicity
- There is no effective antidote for colchicine overdose
Contraindications
- Severe renal impairment (CrCl < 30 mL/min) when combined with CYP3A4 or P-glycoprotein inhibitors
- Severe hepatic impairment combined with renal impairment
- Known hypersensitivity to colchicine
- Blood dyscrasias or significant bone marrow suppression
Drug Interactions
- Strong CYP3A4 inhibitors (clarithromycin, ketoconazole, ritonavir): dramatically increase colchicine levels, potentially fatal — dose reduction or avoidance required
- P-glycoprotein inhibitors (cyclosporine, verapamil): increase colchicine bioavailability significantly
- Statins: increased risk of myopathy and rhabdomyolysis with concurrent use
- Macrolide antibiotics: particularly dangerous interaction leading to colchicine toxicity
- Grapefruit juice may increase colchicine levels
Special Populations
- Renal impairment: dose reduction mandatory; monitor CBC regularly
- Hepatic impairment: dose reduction required; avoid in severe cases
- Elderly: increased susceptibility to toxicity; start with lowest effective dose
- Pregnancy Category C: may cause fetal harm but has been used in FMF patients under close monitoring
Dosage Considerations
- Gout flare: 1.2 mg initially, then 0.6 mg one hour later (max 1.8 mg per course)
- Gout prophylaxis: 0.6 mg once or twice daily
- FMF: 1.2-2.4 mg/day in divided doses
Pharmacokinetic Profile
Colchicine — Pharmacokinetic Curve
SubcutaneousQuick Start
- Typical Dose
- Often initiated at 1.2mg followed by 0.6mg an hour later
Molecular Structure
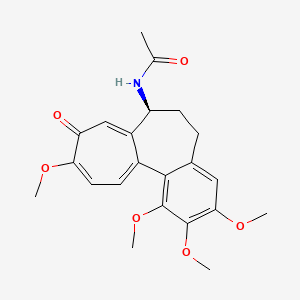
- Formula
- C22H25NO6
- Weight
- 399.4 Da
- PubChem CID
- 6167
- Exact Mass
- 399.1682 Da
- LogP
- 1
- TPSA
- 83.1 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 6
- Rotatable Bonds
- 5
- Complexity
- 740
Identifiers (SMILES, InChI)
InChI=1S/C22H25NO6/c1-12(24)23-16-8-6-13-10-19(27-3)21(28-4)22(29-5)20(13)14-7-9-18(26-2)17(25)11-15(14)16/h7,9-11,16H,6,8H2,1-5H3,(H,23,24)/t16-/m0/s1
IAKHMKGGTNLKSZ-INIZCTEOSA-NSafety Profile
Common Side Effects
- Gastrointestinal Distress:: Common reports of diarrhea, nausea, and abdominal pain
- Drug Interactions:: Dangerous interactions with CYP3A4 inhibitors like Quercetin or Grapefruit
- Toxicity:: Overdose can lead to multi-organ failure due to its systemic effect on cell division
References (2)
- [1]Colchicine --- update on mechanisms of action and therapeutic uses
→ This review details how colchicine inhibits neutrophil chemotaxis and the NLRP3 inflammasome, providing a broad mechanism for treating various systemic inflammatory conditions.
- [2]Cardiovascular effects and safety of long-term colchicine treatment: Cochrane review and meta-analysis
→ A meta-analysis of 39 trials found that while colchicine did not affect all-cause mortality, it significantly reduced cardiovascular mortality in several analytical models.
Cocoa Flavanols
Cocoa flavanols are a group of bioactive plant compounds, specifically flavanones and proanthocyanidins, that enhance nitric oxide bioavailability and improve v
Colivelin
Colivelin is a synthetic chimeric peptide created by fusing the activity-dependent neurotrophic factor-9 (ADNF-9, also called SAL) sequence with a humanin deriv