Sodium Bicarbonate
A well-established alkaline buffering agent used as an ergogenic aid to counteract exercise-induced metabolic acidosis, improving high-intensity exercise performance and delaying fatigue through extracellular pH regulation.
Overview
Sodium bicarbonate (NaHCO₃), commonly known as baking soda, is one of the most extensively researched ergogenic aids in sports science. Its performance-enhancing mechanism is straightforward: during high-intensity anaerobic exercise, rapid glycolysis produces lactate and hydrogen ions (H⁺) that accumulate intracellularly, lowering muscle pH and contributing to fatigue, impaired calcium handling, and reduced glycolytic enzyme activity. Sodium bicarbonate ingestion raises blood bicarbonate concentration and pH (inducing mild metabolic alkalosis), increasing the extracellular buffering capacity and enhancing the efflux of H⁺ ions from working muscle cells via monocarboxylate transporters and the sodium-hydrogen exchanger. This delays the onset of intracellular acidosis and extends time to fatigue.
Meta-analyses consistently demonstrate that sodium bicarbonate supplementation (typically 0.2-0.3 g/kg body weight taken 60-120 minutes before exercise) improves performance in high-intensity activities lasting 1-7 minutes by approximately 2-3%. Benefits are most robust in repeated sprint protocols, 400m-1500m running, rowing, swimming events of 100-400m, and high-intensity interval training. The ergogenic effect extends to resistance exercise, where sodium bicarbonate may increase total training volume at high intensities. These performance improvements, while seemingly modest in percentage terms, are highly meaningful in competitive sport where margins of victory are razor-thin. Sodium bicarbonate also shows promise in team sports requiring repeated high-intensity efforts such as judo, boxing, and CrossFit-style competitions.
The primary limitation of sodium bicarbonate supplementation is gastrointestinal distress, which affects 30-50% of users and can include nausea, bloating, abdominal cramping, and diarrhea. Strategies to mitigate GI issues include serial loading protocols (smaller doses over 3-5 days), taking the dose with a carbohydrate-rich meal, using enteric-coated capsules, or splitting the acute dose into multiple smaller servings over 30-60 minutes. Beta-alanine provides complementary intracellular buffering through carnosine synthesis, and the two are often stacked for additive pH-buffering effects across both intracellular and extracellular compartments. Sodium bicarbonate is not banned by WADA and remains one of the most cost-effective legal ergogenic supplements available.
Mechanism of Action
Sodium bicarbonate (NaHCO3) is the most widely used alkalinizing agent, functioning through the body's principal extracellular buffer system. Upon administration, it dissociates completely into sodium (Na+) and bicarbonate (HCO3-) ions. The bicarbonate ion directly neutralizes excess hydrogen ions (H+) in a simple acid-base reaction: HCO3- + H+ -> H2CO3 (carbonic acid). Carbonic anhydrase then rapidly catalyzes the dehydration of carbonic acid to CO2 and H2O. The generated CO2 is eliminated through pulmonary ventilation, effectively removing the acid load from the body. This reaction shifts the Henderson-Hasselbalch equilibrium toward a higher pH.
In the kidneys, administered bicarbonate increases the filtered bicarbonate load. Under normal conditions, approximately 80-90% of filtered bicarbonate is reabsorbed in the proximal tubule via Na+/H+ exchangers (NHE3) and basolateral Na+/HCO3- cotransporters (NBCe1). When plasma bicarbonate exceeds the renal threshold (approximately 24-26 mEq/L), excess bicarbonate is excreted in urine, producing urinary alkalinization. This urinary alkalinization is therapeutically exploited to enhance excretion of weak acids (e.g., salicylate, methotrexate) through ion trapping and to prevent uric acid and cystine crystallization.
At the cellular level, correction of acidosis by sodium bicarbonate restores optimal pH for enzymatic function, improves cardiac contractility (which is depressed in severe acidosis), and reverses acidosis-induced vasodilation. In exercise physiology, sodium bicarbonate pre-loading increases extracellular buffering capacity, enhancing the efflux of H+ and lactate from exercising muscle via monocarboxylate transporters (MCTs), thereby delaying the onset of intracellular acidosis and muscular fatigue.
Research
Reported Effects
Exercise Duration:: Most effective for activities lasting 30 seconds to 12 minutes, with diminishing returns for longer duration efforts. Individual Response:: Effectiveness varies significantly based on timing relative to peak alkalosis (60-180 minutes pre-exercise), which differs between individuals. Dose-Response:: Clear dose-dependent effects with 0.3 g/kg appearing optimal for single-dose protocols; higher doses don't provide additional benefits. Delivery Method:: Enteric-coated capsules reduce gastrointestinal distress compared to gelatin capsules or powder while maintaining efficacy
- Most effective for activities lasting 30 seconds to 12 minutes, with diminishing returns for longer duration efforts
- Effectiveness varies significantly based on timing relative to peak alkalosis (60-180 minutes pre-exercise), which differs between individuals
- Clear dose-dependent effects with 0.3 g/kg appearing optimal for single-dose protocols; higher doses don't provide additional benefits
- Enteric-coated capsules reduce gastrointestinal distress compared to gelatin capsules or powder while maintaining efficacy
Safety Profile
Safety Profile: Sodium Bicarbonate
Common Side Effects
- Gastrointestinal symptoms: bloating, flatulence, belching (CO2 production upon reaction with stomach acid), abdominal distension, and nausea
- Increased thirst due to sodium load
- Mild peripheral edema (water retention from sodium)
- Frequent urination (alkaline diuresis)
- Altered taste (salty, alkaline)
Serious Adverse Effects
- Metabolic alkalosis: excessive intake raises blood pH above 7.45; symptoms include confusion, muscle twitching, hand tremor, nausea, and in severe cases cardiac arrhythmias and seizures; risk increases with renal impairment
- Sodium overload: each gram contains ~274 mg sodium; chronic high intake can cause hypertension, fluid retention, and heart failure exacerbation; a teaspoon (~4.6 g) contains ~1260 mg sodium
- Hypokalemia: alkalosis drives potassium into cells; dangerously low serum potassium can cause cardiac arrhythmias and muscle weakness
- Hypocalcemia: alkalosis increases protein binding of calcium, reducing ionized calcium; symptoms include paresthesias, tetany, and seizures
- Gastric rupture: extremely rare but reported; rapid CO2 generation from large doses on a full stomach with impaired gastric outlet can cause gastric distension and perforation
- Milk-alkali syndrome: combining sodium bicarbonate with high calcium intake (dairy or supplements) can cause hypercalcemia, renal insufficiency, and metabolic alkalosis
Contraindications
- Metabolic or respiratory alkalosis
- Hypocalcemia (bicarbonate will worsen ionized calcium reduction)
- Hypokalemia (alkalosis exacerbates potassium shifts)
- Severe heart failure or sodium-restricted diets
- Severe renal insufficiency (inability to excrete bicarbonate load)
- GI obstruction or perforation risk
- Concurrent high-calcium intake (milk-alkali syndrome risk)
Drug Interactions
- Virtually all oral medications: sodium bicarbonate raises gastric pH, which affects dissolution, solubility, and absorption of many drugs; separate by 1–2 hours
- Aspirin and salicylates: alkaline urine increases salicylate excretion; may reduce aspirin efficacy
- Lithium: alkaline urine increases lithium clearance; may reduce therapeutic levels
- Methotrexate: urinary alkalinization increases methotrexate excretion; used therapeutically in high-dose MTX protocols but can reduce efficacy of standard dosing
- Tetracyclines and fluoroquinolones: reduced absorption in alkaline gastric environment
- Ketoconazole and itraconazole: require acidic gastric pH for absorption; efficacy reduced by bicarbonate
- Amphetamines and pseudoephedrine: alkaline urine reduces excretion; may increase drug effects and toxicity
- Enteric-coated medications: premature dissolution possible if gastric pH rises above enteric coating threshold
Population-Specific Considerations
- Athletes: 200–300 mg/kg taken 60–90 minutes before exercise is an evidence-based ergogenic aid for high-intensity performance; GI distress is the primary limiting factor; serial dosing protocols may improve tolerability
- Antacid use: FDA-approved OTC antacid; use for occasional heartburn only; chronic daily use risks metabolic alkalosis and sodium overload
- Heart failure / hypertension: sodium content makes this a poor antacid choice; use alternative agents (calcium carbonate, PPIs, H2 blockers)
- Elderly: heightened risk of metabolic alkalosis, hypokalemia, and sodium overload; renal function decline compounds risks
- Pregnancy: occasional antacid use is acceptable; chronic high-dose use risks metabolic alkalosis affecting fetal acid-base balance
- Kidney disease: severely impaired bicarbonate excretion; use only under nephrologist supervision; paradoxically, controlled low-dose bicarbonate supplementation is used therapeutically to manage metabolic acidosis in CKD
Pharmacokinetic Profile
Molecular Structure
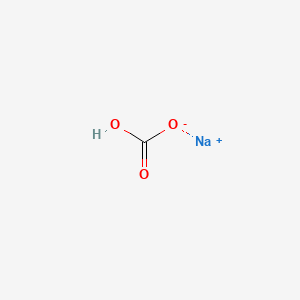
- Formula
- CHNaO3
- Weight
- 84.007 Da
- PubChem CID
- 516892
- Exact Mass
- 83.9823 Da
- TPSA
- 60.4 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 3
- Rotatable Bonds
- 0
- Complexity
- 33
Identifiers (SMILES, InChI)
InChI=1S/CH2O3.Na/c2-1(3)4;/h(H2,2,3,4);/q;+1/p-1
UIIMBOGNXHQVGW-UHFFFAOYSA-MSafety Profile
Common Side Effects
- Gastrointestinal Distress:: Most commonly reported issue including bloating, cramping, nausea, and diarrhea, particularly at higher doses or with rapid consumption
- Individual Variability:: GI symptoms vary significantly between individuals, with some tolerating powder while others require enteric capsules
- Timing Sensitivity:: Taking on empty stomach or too close to exercise increases likelihood of stomach upset and performance impairment
- Dose-Dependent:: Side effects increase substantially above 0.3 g/kg, with minimal additional performance benefit from higher doses
References (4)
- [1]International Society of Sports Nutrition Position Stand: Sodium Bicarbonate and Exercise Performance
→ Comprehensive review confirming that 0.2-0.5 g/kg sodium bicarbonate improves performance in muscular endurance, combat sports, and high-intensity activities lasting 30 seconds to 12 minutes. Optimal single dose is 0.3 g/kg taken 60-180 minutes before exercise.
- [4]Effects of Acute and Multi-Day Low-Dose Sodium Bicarbonate Intake on High-Intensity Endurance Exercise Performance
→ Research comparing acute versus multi-day protocols found that both 0.2 g/kg acute and 0.2 g/kg/day for four days increased blood bicarbonate concentrations, with multi-day supplementation showing trends toward improved time-to-exhaustion.
- [2]Effects of Sodium Bicarbonate Supplementation on Exercise Performance: An Umbrella Review
→ Meta-analysis of systematic reviews showing sodium bicarbonate significantly improves performance in single and multiple-bout high-intensity exercise across various sports and activities in both men and women.
- [3]Sodium Bicarbonate Ingestion in a Fasted State Improves 16.1-km Cycling Time Trial Performance
→ Study found that 0.3 g/kg sodium bicarbonate in both gelatin and enteric capsules improved 16.1-km time trial performance by approximately 34-40 seconds compared to placebo, with enteric capsules causing fewer gastrointestinal side effects.
Sodium Ascorbate
Sodium Ascorbate is the sodium salt of ascorbic acid (vitamin C), a water-soluble antioxidant essential for collagen synthesis, immune function, and iron absorp
Sodium Butyrate
Sodium butyrate is the salt form of butyric acid, a short-chain fatty acid (SCFA) produced by the gut microbiota that acts as a potent histone deacetylase (HDAC