SOMATOSTATIN
Somatostatin (SRIF, somatotropin release-inhibiting factor) is a cyclic tetradecapeptide hormone that inhibits growth hormone secretion, regulates gastrointestinal motility and secretion, and modulates neuroendocrine function through five receptor subtypes (SSTR1-5). It serves as the parent molecule for clinically important analogs including octreotide and lanreotide.
Overview
Somatostatin was identified in 1973 by Brazeau, Vale, and Guillemin during attempts to isolate hypothalamic growth hormone-releasing factor. Instead, they discovered a 14-amino acid cyclic peptide that potently inhibited GH release from anterior pituitary cells (Brazeau et al., 1973). The discovery earned Guillemin a share of the 1977 Nobel Prize in Physiology or Medicine.
Somatostatin's biological role extends far beyond GH regulation. It is a universal "off switch" that inhibits the secretion of numerous hormones (GH, TSH, insulin, glucagon, gastrin, secretin, cholecystokinin, VIP), suppresses exocrine secretion (gastric acid, pancreatic enzymes, bile), reduces GI motility and splanchnic blood flow, and modulates neurotransmission and immune function. These actions are mediated through five G protein-coupled receptor subtypes (SSTR1-5) with distinct tissue distributions and signaling profiles.
The native peptide's clinical utility is severely limited by its ~1-3 minute plasma half-life, which necessitates continuous IV infusion. This limitation motivated the development of metabolically stable analogs: octreotide (SST-14 pharmacophore in an 8-amino acid scaffold) and lanreotide, which retain key biological activities with half-lives measured in hours to weeks.
Mechanism of Action
Somatostatin acts through five receptor subtypes with distinct signaling:
- SSTR1-5 (Gi/Go-coupled GPCRs): All five somatostatin receptors inhibit adenylyl cyclase, reducing cAMP and suppressing hormone secretion. Additional signaling includes activation of phosphotyrosine phosphatases (SHP-1, SHP-2), modulation of MAPK pathways, and activation of inward-rectifying potassium channels (Patel, 1999).
- Growth hormone inhibition: Hypothalamic somatostatin neurons project to the median eminence and release SST-14 into the hypophyseal portal circulation, directly suppressing GH release from somatotroph cells via SSTR2 and SSTR5.
- GI secretion and motility: Somatostatin released from D cells in the gastric antrum and intestinal mucosa inhibits gastrin, gastric acid, pepsin, pancreatic enzymes, and bile secretion. It reduces splanchnic blood flow and GI motility through local paracrine and endocrine actions.
- Pancreatic regulation: Delta cell somatostatin inhibits both insulin (beta cells) and glucagon (alpha cells) secretion in a paracrine manner, serving as a local brake on islet hormone release.
- Antiproliferative effects: SSTR2 and SSTR5 activation inhibits cell proliferation through phosphotyrosine phosphatase activation and cell cycle arrest, forming the basis for somatostatin analog therapy in neuroendocrine tumors (Theodoropoulou & Stalla, 2013).
Research
Glucagonoma
Glucagonomas present with the characteristic necrolytic migratory erythema, diabetes mellitus, weight loss, and venous thromboembolism. Octreotide effectively controls glucagon hypersecretion and resolves the skin rash in most patients, though tumor reduction typically requires surgery or cytotoxic therapy. SSTR2 expression on glucagonoma cells mediates the suppressive effect on glucagon release.
Neuroendocrine Tumors — CLARINET Trial
The CLARINET trial extended the antiproliferative paradigm to lanreotide Autogel and to pancreatic NETs. This phase 3 randomized, double-blind, placebo-controlled trial enrolled 204 patients with nonfunctional, well-differentiated (Ki-67 <10%) GEP-NETs (including pancreatic, midgut, hindgut, and unknown primary). Lanreotide Autogel 120 mg every 28 days significantly prolonged progression-free survival compared to placebo (median not reached vs 18.0 months, HR 0.47, p<0.001). The benefit was consistent across tumor origin (pancreatic and intestinal) and hepatic tumor burden subgroups. CLARINET established lanreotide as first-line antiproliferative therapy for advanced GEP-NETs, regardless of functional status. Caplin, M. E. et al. (2014) — N. Engl. J. Med.
Carcinoid Syndrome
Somatostatin analogs remain the cornerstone treatment for carcinoid syndrome, characterized by flushing, diarrhea, bronchoconstriction, and right heart valve fibrosis caused by serotonin hypersecretion from metastatic midgut NETs. Octreotide LAR controls flushing in approximately 70-80% and diarrhea in 60-75% of patients. For refractory carcinoid syndrome, the TELESTAR trial demonstrated that telotristat ethyl (a tryptophan hydroxylase inhibitor) added to somatostatin analogs further reduced bowel movement frequency, representing the first approved add-on therapy. Kulke, M. H. et al. (2017) — J. Clin. Oncol.
VIPoma (Watery Diarrhea Syndrome)
VIPomas are rare pancreatic neuroendocrine tumors secreting vasoactive intestinal peptide (VIP), causing massive secretory diarrhea (Verner-Morrison syndrome), hypokalemia, and achlorhydria. Octreotide is dramatically effective, reducing diarrheal volume by >50% in most patients within 24-48 hours through suppression of VIP secretion and direct anti-secretory effects on intestinal epithelium. Somatostatin analogs bridge patients to curative surgery or provide long-term symptom control in metastatic disease. O'Dorisio, T. M. et al. (1989) — Metabolism
Cushing's Disease (Pasireotide)
Pasireotide is the only somatostatin analog with efficacy in Cushing's disease, owing to its high SSTR5 affinity. The phase 3 trial randomized 162 patients with Cushing's disease to pasireotide SC 600 μg or 900 μg twice daily. At 6 months, 26.3% of the 900 μg group achieved normalization of urinary free cortisol (UFC ≤ULN), with mean UFC decreasing by approximately 50%. The primary limitation is hyperglycemia, occurring in 73% of patients due to suppression of insulin and incretin secretion via SSTR5 on pancreatic beta cells. Colao, A. et al. (2012) — N. Engl. J. Med. Pasireotide LAR (10 mg or 30 mg monthly IM) was subsequently approved for Cushing's disease with similar efficacy and a more convenient dosing schedule.
Neuroendocrine Tumors — PROMID Trial
The PROMID trial was a landmark phase 3b randomized controlled trial establishing the antiproliferative efficacy of octreotide LAR in well-differentiated midgut neuroendocrine tumors. 85 patients with metastatic midgut NETs (functional and nonfunctional) were randomized to octreotide LAR 30 mg monthly versus placebo. Octreotide LAR significantly prolonged time to tumor progression (14.3 months vs 6.0 months, HR 0.34, p=0.000072). The benefit was most pronounced in patients with low hepatic tumor burden (≤10%) and resected primary tumors. PROMID established that somatostatin analogs have direct antiproliferative effects independent of symptom control. Rinke, A. et al. (2009) — J. Clin. Oncol.
Acromegaly
Somatostatin analogs are first-line medical therapy for acromegaly when surgery fails to achieve biochemical cure (normal IGF-1, GH <1 μg/L). Octreotide LAR and lanreotide Autogel achieve biochemical control (normal IGF-1) in approximately 55-65% of unselected patients, with higher rates in those with moderate GH elevation and tumors expressing high SSTR2 density. The prospective PRIMARYS study demonstrated that lanreotide Autogel 120 mg as primary therapy achieved GH <2.5 μg/L in 63% and normal IGF-1 in 34% of treatment-naive macroadenoma patients after 48 weeks, with significant tumor volume reduction (median -26%). Caron, P. J. et al. (2014) — J. Clin. Endocrinol. Metab. Pasireotide LAR was compared to octreotide LAR in the phase 3 PAOLA study, demonstrating superior biochemical control (15.4% vs 20.5% achieving GH <2.5 and normal IGF-1) in patients inadequately controlled on first-generation analogs, though hyperglycemia was significantly more common with pasireotide. Gadelha, M. R. et al. (2014) — Lancet Diabetes Endocrinol.
GI Bleeding
Intravenous somatostatin infusion (250 mcg/hr) reduces splanchnic blood flow and portal pressure, making it effective for acute variceal bleeding in portal hypertension. Multiple randomized trials have demonstrated efficacy comparable to vasopressin with fewer cardiovascular side effects. Octreotide has largely replaced native somatostatin for this indication due to practical advantages (Moitinho et al., 2001).
Pain Modulation
Somatostatin and its receptors are expressed throughout pain processing pathways, including dorsal root ganglia, spinal cord dorsal horn, and supraspinal pain centers. SSTR2 and SSTR4 activation produces antinociceptive effects in inflammatory and neuropathic pain models. Peripheral somatostatin release from sensory nerve endings contributes to local anti-inflammatory and analgesic effects. Intrathecal somatostatin has been explored for chronic pain, though narrow therapeutic margins limit clinical application (Carlton et al., 2003).
Somatostatin in Diabetes
Somatostatin's dual inhibition of insulin and glucagon has complex implications for glucose homeostasis. SSTR2 antagonism has been explored as a strategy to enhance glucagon counter-regulation in type 1 diabetes, potentially preventing hypoglycemia. Conversely, somatostatin analogs can impair glucose tolerance by suppressing insulin secretion (Kailey et al., 2012).
Neuroendocrine Tumors
Somatostatin receptor expression is a hallmark of neuroendocrine tumors (NETs), making both diagnostic imaging (⁶⁸Ga-DOTATATE PET) and therapy (peptide receptor radionuclide therapy, PRRT) possible. Understanding of SSTR subtype expression patterns in different NET types guides analog selection. The PROMID and CLARINET trials demonstrated that somatostatin analogs (octreotide LAR, lanreotide) exert antiproliferative effects in well-differentiated NETs, extending progression-free survival (Rinke et al., 2009; Caplin et al., 2014).
Safety Profile
Native somatostatin administered by IV infusion is generally well tolerated acutely. Side effects include nausea, abdominal discomfort, and transient hyperglycemia (from insulin suppression). The primary limitation is pharmacokinetic — the 1-3 minute half-life requires continuous infusion, and rebound hormone hypersecretion can occur upon discontinuation. Chronic effects are better characterized with somatostatin analogs (octreotide, lanreotide), where gallstone formation (reduced gallbladder contractility), glucose intolerance, GI side effects (diarrhea, steatorrhea), and injection site reactions are well-documented class effects. Vitamin B12 deficiency can occur with long-term analog therapy.
Pharmacokinetic Profile
SOMATOSTATIN — Pharmacokinetic Curve
Intravenous infusion (native peptide)Quick Start
- Route
- Intravenous infusion (native peptide)
Molecular Structure
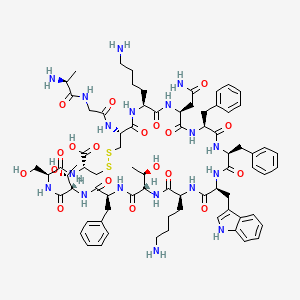
- Formula
- C76H104N18O19S2
- Weight
- 1637.9 Da
- CAS
- 38916-34-6
- PubChem CID
- 16129706
- Exact Mass
- 1636.7167 Da
- LogP
- -3.1
- TPSA
- 664 Ų
- H-Bond Donors
- 22
- H-Bond Acceptors
- 24
- Rotatable Bonds
- 26
- Complexity
- 3240
Identifiers (SMILES, InChI)
InChI=1S/C76H104N18O19S2/c1-41(79)64(100)82-37-61(99)83-58-39-114-115-40-59(76(112)113)92-72(108)57(38-95)91-75(111)63(43(3)97)94-71(107)54(33-46-23-11-6-12-24-46)90-74(110)62(42(2)96)93-66(102)51(28-16-18-30-78)84-69(105)55(34-47-36-81-49-26-14-13-25-48(47)49)88-68(104)53(32-45-21-9-5-10-22-45)86-67(103)52(31-44-19-7-4-8-20-44)87-70(106)56(35-60(80)98)89-65(101)50(85-73(58)109)27-15-17-29-77/h4-14,19-26,36,41-43,50-59,62-63,81,95-97H,15-18,27-35,37-40,77-79H2,1-3H3,(H2,80,98)(H,82,100)(H,83,99)(H,84,105)(H,85,109)(H,86,103)(H,87,106)(H,88,104)(H,89,101)(H,90,110)(H,91,111)(H,92,108)(H,93,102)(H,94,107)(H,112,113)/t41-,42+,43+,50-,51-,52-,53-,54-,55-,56-,57-,58-,59-,62-,63-/m0/s1
NHXLMOGPVYXJNR-ATOGVRKGSA-NResearch Indications
Endocrine
First-generation analogs (octreotide LAR, lanreotide Autogel) normalize IGF-1 in 50-70% of patients. Pasireotide achieves biochemical control in an additional 15-20% who are refractory to first-generation analogs.
Octreotide and lanreotide normalize TSH and thyroid hormones in >90% of thyrotropinomas, with tumor shrinkage in approximately 50% of cases.
Octreotide LAR and lanreotide control flushing and diarrhea in 50-70% of patients with carcinoid syndrome by inhibiting serotonin and other vasoactive peptide release from neuroendocrine tumors.
Oncology
PROMID and CLARINET trials demonstrated that octreotide LAR and lanreotide significantly prolong progression-free survival in well-differentiated GEP-NETs, establishing antiproliferative effect independent of symptom control.
Somatostatin analogs form the basis of peptide receptor radionuclide therapy (PRRT) using 177Lu-DOTATATE (Lutathera), FDA-approved for SSTR-positive GEP-NETs based on NETTER-1 trial.
Some studies suggest octreotide may slow progression in advanced HCC expressing somatostatin receptors, but results are inconsistent and not standard of care.
Gastrointestinal
Octreotide infusion reduces portal venous pressure and is used as adjunct therapy for acute esophageal variceal hemorrhage, comparable to terlipressin in efficacy.
Octreotide effectively manages high-output secretory diarrhea from VIPomas, short bowel syndrome, and chemotherapy-induced diarrhea by inhibiting intestinal secretion.
Somatostatin analogs reduce pancreatic exocrine secretion and are used to manage pancreatic fistulae post-surgery, though evidence for prevention is mixed.
Research Protocols
intravenous Injection
GI Bleeding Intravenous somatostatin infusion (250 mcg/hr) reduces splanchnic blood flow and portal pressure, making it effective for acute variceal bleeding in portal hypertension.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Infusion | 250 mcg | Per protocol | — |
intrathecal Injection
Intrathecal somatostatin has been explored for chronic pain, though narrow therapeutic margins limit clinical application (Carlton et al., 2003).
subcutaneous Injection
Lanreotide (Somatuline) followed with its Autogel depot formulation enabling deep subcutaneous self-injection. Injection site reactions are common with subcutaneous formulations; octreotide LAR intramuscular injection may cause injection site pain and nodule formation.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Octreotide LAR (acromegaly) | 20 mg, 30 mg, 40 mg, 1.0 μg | Per protocol | 28 days |
| Lanreotide Autogel (GEP-NETs, CLARINET protocol) | 120 mg | Per protocol | 28 days(Route: Subcutaneous Injection) |
| Octreotide LAR (PROMID protocol) | 30 mg | Per protocol | 3 months |
| Pasireotide SC (Cushing's disease) | 600 μg, 900 μg | Twice daily | 2 months |
| Carcinoid crisis prevention | 250-500 μg, 50-200 μg | Per protocol | — |
intramuscular Injection
Injection site reactions are common with subcutaneous formulations; octreotide LAR intramuscular injection may cause injection site pain and nodule formation.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Analogs are first-line medical therapy fo | 1 μg | Per protocol | — |
| General Research Protocol | 120 mg, 2.5 μg | Per protocol | 48 weeks |
| General Research Protocol | 30 mg | Monthly | — |
| General Research Protocol | 120 mg | Per protocol | 0 months |
Interactions
Peptide Interactions
Somatostatin inhibits both insulin and glucagon secretion from pancreatic islets. Exogenous insulin combined with somatostatin analogs can produce unpredictable glycemic effects, as glucagon counter-regulation is also suppressed. Blood glucose monitoring is essential to avoid hypoglycemia. (Gerich, 1981, Metabolism)
What to Expect
What to Expect
Rapid onset expected; half-life of ~1-3 minutes (plasma) indicates fast-acting pharmacokinetics
Due to short half-life (~1-3 minutes (plasma)), effects are expected per-dose; consistent daily administration maintains therapeutic levels
Regular administration schedule required; effects are dose-dependent and do not persist between doses
Safety Profile
Common Side Effects
- Metabolic Effects:: Can affect blood glucose, insulin, and glucagon levels requiring monitoring
- Gastrointestinal Issues:: May cause digestive disturbances due to reduced GI secretions
- Growth Hormone Suppression:: Inhibits growth hormone which could be problematic in certain populations
- Injection Site Reactions:: As an injectable medication, may cause local reactions at administration sites
Quality Indicators
What to look for
- Well-established safety profile
- Multiple peer-reviewed studies available
Caution
- Injection site reactions reported
Frequently Asked Questions
References (18)
- [7]Effects of a single dose of N-Acetyl-5-methoxytryptamine (Melatonin) and resistance exercise on the growth hormone/IGF-1 axis in young males and females
→ The study examined melatonin's effects on growth hormone and somatostatin levels, finding that melatonin and resistance exercise influence the GH/IGF-1 axis and somatostatin secretion.
- [10]
- [11]Bauer W et al SMS 201-995: a very potent and selective octapeptide analogue of somatostatin with prolonged action Life Sci (1982)
- [9]Brazeau P et al Hypothalamic polypeptide that inhibits the secretion of immunoreactive pituitary growth hormone Science (1973)
- [2]Dumping Syndrome: Pragmatic Treatment Options and Experimental Approaches for Improving Clinical Outcomes
→ Somatostatin analogs are effective therapeutic options for managing dumping syndrome, a common complication after gastric and bariatric surgery that significantly impacts quality of life.
- [3]Glucagon and diabetes
→ Somatostatin supplementation can suppress excess glucagon and growth hormone secretion in diabetes management, helping to correct both hyperglycemia and hyperglucagonemia when combined with insulin therapy.
- [4]Esophageal varices
→ Somatostatin is used alongside other treatments to control acute variceal bleeding in patients with liver disease, representing a pharmacological option for this life-threatening condition.
- [5]Malignant insulinoma
→ Somatostatin analogs like octreotide can be used in combination with other therapies for managing neuroendocrine tumors, with octreotide scintigraphy helping identify tumors expressing somatostatin receptors for targeted treatment.
- [6]Tumor-induced osteomalacia: An overview
→ Somatostatin receptor-based PET imaging is a first-line investigation for localizing phosphaturic mesenchymal tumors in tumor-induced osteomalacia, aiding in diagnosis and treatment planning.
- [8]Pharmacological interventions for acute pancreatitis
→ A comprehensive Cochrane review assessed various pharmacological interventions including somatostatin for acute pancreatitis, evaluating the evidence for medical treatments beyond supportive care.
- [13]Caplin ME et al Lanreotide in metastatic enteropancreatic neuroendocrine tumors (CLARINET) N Engl J Med (2014)
- [14]Theodoropoulou M, Stalla GK Somatostatin receptors: from signaling to clinical practice Front Neuroendocrinol (2013)
- [15]Moitinho E et al Prognostic value of early measurements of portal pressure in acute variceal bleeding Gastroenterology (2001)
- [18]
- [12]
- [17]Kailey B et al SSTR2 is the functionally dominant somatostatin receptor in human pancreatic beta- and alpha-cells Am J Physiol Endocrinol Metab (2012)
- [16]Carlton SM et al Somatostatin inhibits the transient receptor potential vanilloid 1 (TRPV1) receptor to reduce peripheral nociception J Pharmacol Exp Ther (2003)
- [1]Somatostatin and octreotide on the treatment of acute pancreatitis - basic and clinical studies for three decades
→ Somatostatin and octreotide show anti-inflammatory effects in acute pancreatitis treatment beyond just reducing pancreatic secretions, with potential benefits in reducing sphincter of Oddi tone and addressing the inflammatory injury associated with the condition.
Solcoseryl
Solcoseryl is a protein-free hemodialysate derived from calf blood containing low-molecular-weight compounds including nucleotides, amino acids, and electrolyte
Somatropin (rHGH)
Somatropin is the recombinant DNA-derived form of human growth hormone (hGH), a 191-amino acid single-chain polypeptide secreted by somatotroph cells of the ant