TOREMIFENE
A selective estrogen receptor modulator (SERM) structurally related to tamoxifen, used clinically for the treatment of estrogen receptor-positive breast cancer and off-label as an anti-estrogen in hormone optimization protocols.
Toremifene is a selective estrogen receptor modulator (SERM) and a chlorinated derivative of tamoxifen, primarily used in the treatment of hormone receptor-positive breast cancer in postmenopausal women. It works by binding to estrogen receptors and exhibiting both antiestrogenic effects in breast tissue and some estrogenic agonist properties in other tissues like bone and cardiovascular system. Unlike tamoxifen, toremifene appears to have a more favorable safety profile regarding endometrial effects while maintaining similar efficacy in breast cancer treatment.
Research
Reported Effects
Breast Cancer Efficacy:: Multiple meta-analyses confirm equivalent or superior effectiveness to tamoxifen for breast cancer treatment with 5-year survival benefits. Endometrial Safety:: Safer endometrial profile than tamoxifen with lower risk of promoting endometrial cancer growth. Metabolic Effects:: More favorable effects on liver health and fatty liver prevention compared to tamoxifen. Long-term Tolerability:: Well-tolerated with rare and mild side effects in clinical trials spanning years of treatment
- Multiple meta-analyses confirm equivalent or superior effectiveness to tamoxifen for breast cancer treatment with 5-year survival benefits
- Safer endometrial profile than tamoxifen with lower risk of promoting endometrial cancer growth
- More favorable effects on liver health and fatty liver prevention compared to tamoxifen
- Well-tolerated with rare and mild side effects in clinical trials spanning years of treatment
Safety Profile
Safety Profile: Toremifene
Common Side Effects
- Hot flashes (the most common side effect, occurring in up to 35% of patients)
- Nausea, vomiting, and gastrointestinal discomfort
- Vaginal discharge, vaginal dryness, and irregular menstruation
- Sweating and flushing
- Peripheral edema and weight changes
- Dizziness and fatigue
- Elevated liver enzymes (transient and usually asymptomatic)
Serious Adverse Effects
- QT prolongation and cardiac arrhythmias: Toremifene carries a risk of QTc prolongation that may lead to torsades de pointes; ECG monitoring recommended, especially in patients with risk factors
- Thromboembolic events: Increased risk of deep vein thrombosis (DVT), pulmonary embolism (PE), and stroke, similar to other selective estrogen receptor modulators (SERMs)
- Endometrial changes: Endometrial hyperplasia, polyps, and rarely endometrial cancer with long-term use
- Hepatotoxicity: Elevated liver enzymes; rare cases of significant hepatic injury
- Hypercalcemia: May occur in patients with bone metastases, particularly during initial treatment
- Ocular effects: Corneal keratopathy, cataracts, and visual disturbances reported with prolonged use
Contraindications
- Known hypersensitivity to toremifene or any excipients
- History of or existing thromboembolic disease (DVT, PE, stroke) unless benefits clearly outweigh risks
- Pre-existing QT prolongation, uncorrected hypokalemia or hypomagnesemia, or concurrent use of QT-prolonging drugs
- Endometrial hyperplasia or unexplained vaginal bleeding
- Severe hepatic impairment
- Pregnancy (FDA Category D) and lactation; may cause fetal harm
Drug Interactions
- QT-prolonging drugs (antiarrhythmics, fluoroquinolones, macrolide antibiotics, antipsychotics): Additive QT prolongation; avoid combination or monitor ECG closely
- CYP3A4 inhibitors (ketoconazole, itraconazole, erythromycin): Increase toremifene plasma levels; heightened risk of QT prolongation
- CYP3A4 inducers (rifampin, phenytoin, carbamazepine): Decrease toremifene levels; may reduce efficacy
- Anticoagulants (warfarin): SERMs may potentiate anticoagulant effects; monitor INR
- Thiazide diuretics: May worsen hypokalemia, increasing QT prolongation risk
Population-Specific Considerations
- Postmenopausal women with breast cancer: Primary indication (metastatic ER+ breast cancer); monitor endometrial thickness and thromboembolic risk
- Elderly: Increased cardiovascular and thromboembolic risk; ECG and electrolyte monitoring essential
- Men (off-label use): Used off-label for gynecomastia prevention during testosterone or anabolic steroid use; monitor liver function and lipids
- Cardiac patients: Avoid in patients with significant cardiac history; obtain baseline ECG and correct electrolyte abnormalities before starting
- Hepatic impairment: Reduced clearance; avoid in severe impairment; monitor liver function in mild-moderate impairment
Pharmacokinetic Profile
Quick Start
- Typical Dose
- 60mg daily administered orally for breast cancer treatment
Molecular Structure
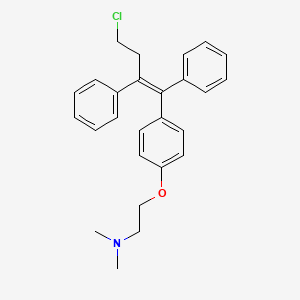
- Formula
- C26H28ClNO
- Weight
- 406.0 Da
- PubChem CID
- 3005573
- Exact Mass
- 405.1859 Da
- LogP
- 7.2
- TPSA
- 12.5 Ų
- H-Bond Donors
- 0
- H-Bond Acceptors
- 2
- Rotatable Bonds
- 9
- Complexity
- 483
Identifiers (SMILES, InChI)
InChI=1S/C26H28ClNO/c1-28(2)19-20-29-24-15-13-23(14-16-24)26(22-11-7-4-8-12-22)25(17-18-27)21-9-5-3-6-10-21/h3-16H,17-20H2,1-2H3/b26-25-
XFCLJVABOIYOMF-QPLCGJKRSA-NSafety Profile
Common Side Effects
- Vasomotor Symptoms:: Hot flashes and sweating commonly reported, similar to other SERMs
- Vaginal Discharge:: More common than with control but generally mild
- Hepatic Effects:: Generally does not negatively affect liver function at therapeutic doses
- Thromboembolic Risk:: Similar low risk of blood clots as tamoxifen, including deep vein thrombosis and pulmonary embolism
References (8)
- [3]Effects of Tamoxifen vs. Toremifene on fatty liver development and lipid profiles in breast Cancer
→ Retrospective analysis of 308 breast cancer patients showed toremifene had more favorable effects on preventing fatty liver development compared to tamoxifen, while both drugs affected lipid profiles differently.
- [4]Comparison of effects of tamoxifen and Toremifene on hepatic function and serum lipids in breast cancer patients during adjuvant endocrine therapy
→ Study of 597 patients found neither toremifene nor tamoxifen negatively affected hepatic function, with toremifene showing similar effects on lipid profiles and potentially stronger cholesterol-lowering effects than tamoxifen.
- [5]Chronic effects of toremifene on the vasculature of menopause-induced rats
→ Four-week treatment with toremifene improved vascular responses to acetylcholine and increased endothelium-dependent relaxation in ovariectomized rats, suggesting beneficial cardiovascular effects similar to estrogen.
- [6]Effects of the antiestrogens tamoxifen, toremifene, and ICI 182,780 on endometrial cancer growth
→ Study in athymic mice showed that unlike tamoxifen, toremifene did not promote endometrial cancer growth and may have a more favorable endometrial safety profile.
- [7]Clinical pharmacokinetics of toremifene
→ Toremifene has approximately 100% oral bioavailability, is highly protein-bound (>95%), metabolized by liver cytochrome P450 enzymes, and has a half-life of approximately 5 days with steady state reached by 6 weeks.
- [8]Combination immunotherapy of the P815 murine mastocytoma with killer cells, IL-2 and anti-estrogens
→ Toremifene enhanced cancer immunotherapy when combined with cytotoxic T lymphocytes and IL-2, increasing cure rates from 25% to 75% in tumor-bearing mice, suggesting immune-modulating properties.
- [1]Effects of toremifene versus tamoxifen on breast cancer patients: a meta-analysis
→ Meta-analysis of 23 trials (7,242 patients) showed toremifene was associated with higher 5-year survival rates in early stage breast cancer compared to tamoxifen, with similar efficacy but different effects on lipid profiles and bone mineral density.
- [2]Toremifene is an effective and safe alternative to tamoxifen in adjuvant endocrine therapy for breast cancer: results of four randomized trials
→ Four randomized trials including 3,747 patients found no significant difference in overall survival or disease-free survival between toremifene and tamoxifen, with similar rates of thromboembolic events, confirming toremifene as an effective alternative.
TOCOTRIENOLS
Tocotrienols are a subfamily of vitamin E compounds that differ from tocopherols by having an unsaturated isoprenoid side chain. They possess potent antioxidant
TRANS-RESVERATROL
Trans-resveratrol is a polyphenolic compound found naturally in grapes, berries, and other plants that acts as a phytoestrogen and activates SIRT1 pathways. It