Hypericin
A naphthodianthrone pigment from St. John's Wort with photodynamic, antiviral, and mood-modulating properties distinct from its companion compound hyperforin.
Overview
Hypericin is a red-pigmented naphthodianthrone compound found in the dark glands of Hypericum perforatum (St. John's Wort) flowers and leaves. While hyperforin is now recognized as the primary antidepressant constituent, hypericin was historically considered the main active compound and remains the standard marker used to characterize St. John's Wort extracts (typically standardized to 0.3% hypericin). Hypericin possesses a distinct pharmacological profile: it inhibits monoamine oxidase (MAO) types A and B at higher concentrations, modulates sigma receptors, binds to NMDA receptors, and demonstrates potent photodynamic properties that have opened therapeutic avenues well beyond mood support.
The photodynamic activity of hypericin is its most scientifically compelling feature. Upon light activation, hypericin generates singlet oxygen and other reactive oxygen species with high quantum yield, making it a potent photosensitizer. This property has been extensively investigated in photodynamic therapy (PDT) for cancer, where hypericin selectively accumulates in tumor tissue and, upon light activation, induces apoptosis through mitochondrial and endoplasmic reticulum stress pathways. Clinical trials have explored hypericin-PDT for bladder cancer (fluorescence cystoscopy and treatment), non-melanoma skin cancers, and glioblastoma. Hypericin also demonstrates broad-spectrum antiviral activity through photodynamic inactivation of enveloped viruses including HIV, herpes simplex, hepatitis C, and influenza.
In the context of mood disorders, hypericin contributes to but does not solely account for St. John's Wort's antidepressant effects. Its MAO inhibition at therapeutic concentrations is relatively mild, and its dopamine beta-hydroxylase inhibition may modulate catecholamine synthesis. The photosensitivity it causes — manifesting as exaggerated sunburn risk — is the most notable side effect of St. John's Wort supplementation, particularly at higher doses or in fair-skinned individuals. Unlike hyperforin, hypericin does not significantly induce cytochrome P450 enzymes, which has led to interest in developing hypericin-enriched, low-hyperforin extracts that could provide antidepressant and antiviral benefits with reduced drug interaction potential. Research continues into hypericin's applications in diagnostic imaging, antimicrobial photodynamic therapy, and as a fluorescent biomarker for cancer detection.
Mechanism of Action
Hypericin is a naturally occurring naphthodianthrone pigment found in St. John's wort (Hypericum perforatum). It exerts antidepressant effects through non-competitive inhibition of the reuptake of multiple neurotransmitters, including serotonin, dopamine, norepinephrine, GABA, and glutamate. By increasing synaptic concentrations of these neurotransmitters, hypericin modulates mood and cognitive function. It also inhibits monoamine oxidase (MAO) and dopamine-beta-hydroxylase, providing additional neurotransmitter-modulating effects.
Hypericin is a potent inhibitor of several protein kinases, notably Protein Kinase C (PKC) and Janus Kinase 1 (JAK1). PKC inhibition contributes to anti-proliferative effects, while JAK1 inhibition underlies anti-inflammatory properties by disrupting cytokine signaling pathways. These kinase-inhibitory activities have generated significant interest in hypericin as a potential anti-cancer and anti-inflammatory agent.
One of hypericin's most distinctive properties is its photodynamic activity. When exposed to visible light, hypericin acts as a potent photosensitizer, absorbing light energy and transferring it to molecular oxygen to generate reactive oxygen species (ROS) including singlet oxygen and superoxide radicals. This photodynamic mechanism induces apoptosis and necrosis in tumor cells and is also the basis for its antiviral activity, as ROS can inactivate enveloped viruses. This property is exploited in photodynamic therapy (PDT) for cancer treatment.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Delayed Onset:: Benefits typically require 2-4 weeks of consistent daily use before noticeable effects appear, similar to prescription antidepressants. Extract Variability:: Effectiveness varies significantly between brands and extract types, with hypericin-standardized (0.3%) and hyperforin-standardized (2%) formulations showing different results. Mild to Moderate Cases:: Most effective for mild to moderate depression and anxiety; users with severe depression report limited benefits. Individual Response:: Highly variable individual responses, with some users experiencing significant benefits while others report minimal or no effects
- Benefits typically require 2-4 weeks of consistent daily use before noticeable effects appear, similar to prescription antidepressants
- Effectiveness varies significantly between brands and extract types, with hypericin-standardized (0.3%) and hyperforin-standardized (2%) formulations showing different results
- Most effective for mild to moderate depression and anxiety; users with severe depression report limited benefits
- Highly variable individual responses, with some users experiencing significant benefits while others report minimal or no effects
Safety Profile
Safety Profile: Hypericin
Common Side Effects
- Photosensitivity (most characteristic): erythema, itching, and tingling of sun-exposed skin, even at standard supplemental doses
- Gastrointestinal complaints: nausea, mild abdominal discomfort, and diarrhea
- Headache and mild dizziness
- Fatigue or restlessness
- Dry mouth
Serious Adverse Effects
- Severe phototoxic dermatitis: at higher doses (>0.5 mg/day pure hypericin), significant risk of blistering, edema, and second-degree burns upon sun exposure
- Phototoxic retinopathy: theoretical risk with prolonged high-dose use and UV exposure; documented in animal models
- Serotonin syndrome: when combined with serotonergic medications, similar to hyperforin but hypericin contributes independently through MAO-A inhibition
- Peripheral neuropathy: rare reports with long-term high-dose use
- Hepatotoxicity: isolated case reports of elevated transaminases
Contraindications
- Known photosensitivity disorders (porphyria, systemic lupus erythematosus, xeroderma pigmentosum)
- Concurrent photodynamic therapy or planned significant UV exposure
- Concurrent MAOIs or serotonergic medications
- Severe hepatic impairment
- Known allergy to Hypericum perforatum or its constituents
Drug Interactions
- Serotonergic drugs (SSRIs, SNRIs, MAOIs, triptans): risk of serotonin syndrome through MAO-A inhibition
- Photosensitizing medications (tetracyclines, fluoroquinolones, thiazides, sulfonamides, amiodarone): additive phototoxicity
- CYP enzyme interactions: weaker CYP inducer than hyperforin, but still may modestly affect CYP3A4 substrates
- Antiretroviral agents: may reduce efficacy of protease inhibitors and NNRTIs
- Anesthetics: discontinue before surgery due to potential MAO-inhibiting effects and photosensitization under surgical lights
Population-Specific Considerations
- Pregnancy / lactation: insufficient safety data; MAO inhibition may affect fetal neurotransmitter development; avoid during pregnancy and breastfeeding
- Children: no established safe dosing; avoid unless under dermatological supervision for specific indications (e.g., photodynamic therapy research)
- Fair-skinned individuals: dramatically higher phototoxicity risk; strict photoprotection required (SPF 50+, protective clothing)
- Outdoor workers / athletes: strongly advise against use unless UV exposure can be rigorously controlled
- Elderly: increased skin fragility amplifies phototoxic reactions; lower doses recommended
Pharmacokinetic Profile
Quick Start
- Typical Dose
- 900mg per day divided into three 300mg doses, standardized to 0.3% hypericin, is the most commonly referenced effective dosage
Molecular Structure
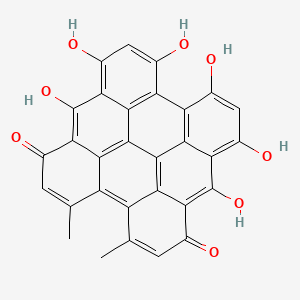
- Formula
- C30H16O8
- Weight
- 504.4 Da
- PubChem CID
- 3663
- Exact Mass
- 504.0845 Da
- LogP
- 5.7
- TPSA
- 156 Ų
- H-Bond Donors
- 6
- H-Bond Acceptors
- 8
- Rotatable Bonds
- 0
- Complexity
- 1090
Identifiers (SMILES, InChI)
InChI=1S/C30H16O8/c1-7-3-9(31)19-23-15(7)16-8(2)4-10(32)20-24(16)28-26-18(12(34)6-14(36)22(26)30(20)38)17-11(33)5-13(35)21(29(19)37)25(17)27(23)28/h3-6,33-38H,1-2H3
YDOIFHVUBCIUHF-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Photosensitivity and Eye Concerns:: Major worry about hypericin causing retinal damage or cataracts with light exposure, though risk with hyperforin formulations appears lower
- Drug Interactions:: Significant interactions with medications including SSRIs, birth control, and various prescription drugs due to effects on liver enzymes
- Digestive Issues:: Some users report appetite changes, nausea, or gastrointestinal discomfort, particularly when starting
- Cognitive Effects:: Reports of brain fog, restlessness, impatience, or difficulty focusing in some users, especially in early weeks
References (3)
- [1]Effect of Hypericum perforatum (St John's Wort) in Major Depressive Disorder
→ Clinical trial by the Hypericum Depression Trial Study Group examining the efficacy of St John's Wort extract for treating major depression, comparing it to placebo and standard antidepressants.
- [2]St John's wort (Hypericum perforatum L.): a review of its chemistry, pharmacology and clinical properties
→ Comprehensive review of hypericin and hyperforin chemistry, mechanisms of action including MAO inhibition and receptor interactions, and clinical applications for depression treatment.
- [3]St. John's wort (Hypericum perforatum): a review of the current pharmacological, toxicological, and clinical literature
→ Extensive review covering pharmacological mechanisms, potential toxicity concerns including photosensitivity, and clinical efficacy data for depression and anxiety disorders.
Hyperforin
Hyperforin is a phloroglucinol derivative and principal active compound in St. John's Wort (Hypericum perforatum) responsible for its antidepressant effects. It
I3C
Indole-3-carbinol (I3C) is a naturally occurring compound found in cruciferous vegetables like broccoli and Brussels sprouts that is converted to DIM (diindolyl