Rutin
A flavonoid glycoside (quercetin-3-O-rutinoside) found in buckwheat, citrus, and numerous plants that strengthens capillary walls, reduces vascular permeability, and provides anti-inflammatory and antioxidant protection with established use in vascular health.
Overview
Rutin (quercetin-3-O-rutinoside) is a flavonol glycoside composed of the aglycone quercetin bound to the disaccharide rutinose (rhamnose-glucose). Named after Ruta graveolens (common rue) from which it was first isolated, rutin is one of the most abundant and widely distributed flavonoids in the plant kingdom, found in buckwheat (its richest dietary source), citrus fruits (particularly the peel and pith), asparagus, green tea, elderflower, and numerous medicinal herbs. Rutin has been used pharmaceutically since the mid-20th century, when Albert Szent-Gyorgyi (who also discovered vitamin C) identified it as a component of "vitamin P" — a term originally proposed for factors that reduced capillary fragility and permeability.
Rutin's best-established therapeutic application is vascular health. It strengthens capillary walls by cross-linking collagen fibers in the vascular basement membrane, reduces endothelial permeability through inhibition of VEGF signaling, and decreases platelet aggregation by modulating phospholipase C and intracellular calcium mobilization. These properties underlie its clinical use in chronic venous insufficiency (CVI), varicose veins, hemorrhoids, and lymphedema, where controlled trials have demonstrated reductions in leg swelling, heaviness, pain, and capillary fragility. Hydroxyethylrutosides (semi-synthetic rutin derivatives) are approved pharmaceutical agents for CVI in many European countries. Rutin also inhibits LDL oxidation, modulates endothelial nitric oxide production, and reduces arterial stiffness, contributing to broader cardiovascular protection that complements hesperidin and other citrus bioflavonoids.
Beyond vascular effects, rutin provides significant anti-inflammatory and antioxidant activity. It inhibits NF-kB, suppresses COX-2 and iNOS, chelates pro-oxidant iron and copper ions, and scavenges superoxide and hydroxyl radicals. Rutin demonstrates neuroprotective properties — protecting against ischemia/reperfusion injury and neurotoxin-induced cell death in animal models — and has shown anti-diabetic effects through alpha-glucosidase inhibition and improvement of insulin sensitivity. A notable pharmacological distinction from its aglycone quercetin is rutin's superior water solubility and gentler gastrointestinal profile, though its oral bioavailability depends on gut microbial hydrolysis to liberate quercetin for absorption. This makes gut microbiome composition an important determinant of rutin's efficacy. Typical supplemental doses range from 250-1,000 mg/day. Rutin pairs well with vitamin C (they are traditionally co-supplemented for capillary health), diosmin, and horse chestnut extract for comprehensive venous support.
Mechanism of Action
Rutin (rutoside, quercetin-3-O-rutinoside) is a flavonol glycoside consisting of quercetin bound to the disaccharide rutinose. Its primary pharmacological action is venotonic and capillary-stabilizing. Rutin strengthens blood vessel walls by interacting with vascular endothelial cell membranes and modulating collagen cross-linking, reducing capillary permeability and fragility. It inhibits hyaluronidase, the enzyme that degrades hyaluronic acid in the extracellular matrix, thereby preserving the structural integrity of the capillary basement membrane. These properties underpin its clinical use in chronic venous insufficiency and hemorrhoidal disease.
As an antioxidant, rutin operates through multiple mechanisms. Its catechol (ortho-dihydroxy) group on the B-ring enables direct scavenging of superoxide, hydroxyl, and peroxyl radicals. It chelates transition metal ions (Fe2+ and Cu2+) that would otherwise catalyze Fenton reactions generating hydroxyl radicals. Rutin also inhibits xanthine oxidase and NADPH oxidase, reducing endogenous superoxide production. In vivo, rutin is metabolized by gut microbiota to quercetin and other phenolic metabolites, which contribute additional antioxidant and anti-inflammatory effects.
Rutin's anti-inflammatory mechanism involves inhibition of phospholipase A2 (PLA2), reducing arachidonic acid release from membrane phospholipids. It also directly inhibits COX-2 and 5-lipoxygenase, suppressing prostaglandin and leukotriene synthesis. Through NF-kB pathway inhibition, rutin reduces expression of TNF-alpha, IL-1beta, IL-6, and adhesion molecules (ICAM-1, VCAM-1). Its antiplatelet effects involve inhibition of platelet-activating factor (PAF) and thromboxane A2, contributing to its cardiovascular protective profile.
Research
Reported Effects
Hemorrhoid Treatment:: Effectiveness varies widely; some see no benefit at 500-1000mg daily for a week, while others report success with higher doses (2000mg+) combined with bioflavonoids. Vascular Benefits:: Shows consistent positive results for circulatory issues when combined with nattokinase, serrapeptase, or other vascular support compounds. Dose-Response Relationship:: Higher doses (2000-3000mg) appear more effective than standard 500mg doses, particularly for acute conditions. Synergistic Effects:: Works better when combined with vitamin C, diosmin/hesperidin, or other bioflavonoids rather than as standalone supplement
- Effectiveness varies widely; some see no benefit at 500-1000mg daily for a week, while others report success with higher doses (2000mg+) combined with bioflavonoids
- Shows consistent positive results for circulatory issues when combined with nattokinase, serrapeptase, or other vascular support compounds
- Higher doses (2000-3000mg) appear more effective than standard 500mg doses, particularly for acute conditions
- Works better when combined with vitamin C, diosmin/hesperidin, or other bioflavonoids rather than as standalone supplement
Safety Profile
Safety Profile: Rutin
Common Side Effects
- Gastrointestinal symptoms: nausea, bloating, diarrhea, and stomach discomfort
- Headache and mild dizziness
- Skin flushing
- Muscle stiffness at higher doses
- Blurred vision (rare)
Serious Adverse Effects
- Rare allergic reactions including urticaria and anaphylaxis
- Theoretical risk of kidney damage at very high doses (rutin metabolites include oxalate)
- Potential for excessive bleeding due to antiplatelet and capillary-stabilizing effects at high doses
- Rare reports of thyroid function alteration in animal models
Contraindications
- Known allergy to rutin or related bioflavonoids (quercetin, hesperidin)
- Active bleeding disorders
- Pre-surgical period (discontinue 2 weeks before surgery)
- Severe renal impairment (oxalate metabolite concern)
Drug Interactions
- Anticoagulants and antiplatelets: Additive bleeding risk
- Quinolone antibiotics (ciprofloxacin, norfloxacin): Rutin may reduce absorption of fluoroquinolones
- Iron supplements: May chelate iron and reduce absorption
- Cyclosporine: May alter drug metabolism via CYP3A4 modulation
- Antihypertensives: Mild additive blood pressure–lowering effect
Population-Specific Considerations
- Chronic venous insufficiency: Best-studied indication; standard dose 500–1000 mg/day of troxerutin (semi-synthetic derivative)
- Hemorrhoid patients: Commonly used with good tolerability
- Pregnancy: Historically used in some countries for capillary fragility in pregnancy, but modern safety data insufficient; consult provider
- Elderly: Well tolerated; monitor renal function with long-term high-dose use
Pharmacokinetic Profile
Quick Start
- Typical Dose
- 500mg daily is commonly available but may be insufficient for therapeutic effects in some conditions
Molecular Structure
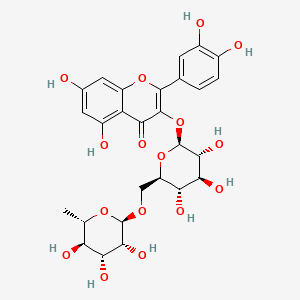
- Formula
- C27H30O16
- Weight
- 610.5 Da
- PubChem CID
- 5280805
- Exact Mass
- 610.1534 Da
- LogP
- -1.3
- TPSA
- 266 Ų
- H-Bond Donors
- 10
- H-Bond Acceptors
- 16
- Rotatable Bonds
- 6
- Complexity
- 1020
Identifiers (SMILES, InChI)
InChI=1S/C27H30O16/c1-8-17(32)20(35)22(37)26(40-8)39-7-15-18(33)21(36)23(38)27(42-15)43-25-19(34)16-13(31)5-10(28)6-14(16)41-24(25)9-2-3-11(29)12(30)4-9/h2-6,8,15,17-18,20-23,26-33,35-38H,7H2,1H3/t8-,15+,17-,18+,20+,21-,22+,23+,26+,27-/m0/s1
IKGXIBQEEMLURG-NVPNHPEKSA-NSafety Profile
Common Side Effects
- Minimal Reported:: Very few users report negative side effects even at high doses (2000mg+)
- Bioavailability Concerns:: Low absorption noted in research, which may limit effectiveness at standard doses
- Gastrointestinal:: No significant digestive issues reported, even with high-dose protocols
- Long-term Safety:: Generally considered safe with users reporting sustained use without adverse effects
References (4)
- [2]The Pharmacological Potential of Rutin
→ Review highlighting rutin's diverse pharmacological effects including antioxidant, anti-inflammatory, anti-apoptotic, and vascular protective properties, with discussion of its clinical applications.
- [4]Molecular mechanisms of neuroprotective effect of rutin
→ Recent review detailing rutin's neuroprotective mechanisms including antioxidant, anti-inflammatory, anti-apoptotic effects, and its role in protecting the blood-brain barrier and enhancing neural function.
- [1]An up-to-date review of rutin and its biological and pharmacological activities
→ Comprehensive review demonstrating rutin's multiple therapeutic properties including antioxidant, anti-inflammatory, neuroprotective, and cardiovascular benefits across various disease models.
- [3]Bioavailability and efficiency of rutin as an antioxidant: a human supplementation study
→ Human study examining rutin's absorption and antioxidant capacity, demonstrating measurable antioxidant effects despite relatively low bioavailability.
Rosmarinic Acid
Rosmarinic acid is a natural polyphenolic compound found in plants of the Lamiaceae and Boraginaceae families, formed from caffeic acid and 3,4-dihydroxyphenyl
S-Acetyl Glutathione
S-Acetyl Glutathione (SAG) is a modified form of glutathione where an acetyl group is attached to the sulfur atom, making it more stable and bioavailable than r