Picamilon
A synthetic compound combining niacin and GABA that crosses the blood-brain barrier, where it is hydrolyzed to release both components, producing vasodilatory and anxiolytic effects used in Russian neurological practice.
Overview
Picamilon (also written as pikamilon) is a synthetic molecule formed by conjugating GABA (gamma-aminobutyric acid) with niacin (nicotinic acid) via a peptide bond. Developed in the Soviet Union in 1969 at the All-Union Vitamin Scientific Research Institute, picamilon was designed to solve a fundamental pharmacological challenge: GABA itself cannot cross the blood-brain barrier (BBB) in meaningful quantities when administered orally, severely limiting its therapeutic utility as a central anxiolytic or neuroprotective agent. By conjugating GABA with the lipophilic niacin moiety, picamilon achieves BBB penetration, after which cerebral esterases hydrolyze the bond to release both GABA and niacin directly into brain tissue.
The dual release of GABA and niacin produces complementary pharmacological effects. GABA acts on GABA-A and GABA-B receptors to produce anxiolytic, mildly sedative, and anticonvulsant activity without the dependency risks associated with benzodiazepines. Niacin, meanwhile, acts as a cerebral vasodilator by stimulating prostaglandin D2 and E2 production in vascular endothelium, improving cerebral blood flow and oxygen delivery. This combination of anxiolysis and cerebrovascular support makes picamilon particularly interesting for conditions involving both anxiety and cerebral hypoperfusion, such as cerebrovascular insufficiency, migraine, and age-related cognitive decline. In Russia, picamilon is an approved prescription drug used for a range of neurological and psychiatric conditions including anxiety disorders, asthenic states, depression, traumatic brain injury sequelae, and glaucoma (where improved ocular blood flow may be beneficial).
In the Western nootropic community, picamilon has been used as a supplement for anxiolytic and cognitive-enhancing purposes, though the FDA issued warning letters to supplement manufacturers in 2015 stating that picamilon does not qualify as a dietary ingredient. Typical dosing in Russian clinical practice ranges from 20-150 mg/day, divided into two or three doses. Picamilon's mechanism is distinct from other GABAergic nootropics like phenibut, which acts as a GABA-B agonist and has a more pronounced sedative and tolerance-building profile. Other nootropics targeting cerebral circulation include vinpocetine and ginkgo biloba.
Mechanism of Action
Picamilon (nicotinoyl-GABA) operates as a prodrug designed to overcome the poor blood-brain barrier (BBB) permeability of GABA. The molecule consists of GABA covalently bonded to nicotinic acid (vitamin B3/niacin) via a peptide bond. This conjugation allows the intact molecule to cross the BBB, likely utilizing the transport mechanisms available to nicotinic acid. Once inside the central nervous system, picamilon is hydrolyzed by amidase enzymes, releasing free GABA and nicotinic acid in the brain parenchyma.
The released GABA can then interact with GABA-A and GABA-B receptors, mediating inhibitory neurotransmission. GABA-A receptor activation opens chloride ion channels, producing rapid inhibitory postsynaptic potentials, while GABA-B receptor activation modulates potassium and calcium channels via G-protein coupling. These actions contribute to anxiolytic, sedative, and anticonvulsant effects. However, a 2023 study in Basic and Clinical Pharmacology and Toxicology found picamilon to be inactive against 50 biological targets in screening assays, suggesting that its direct pharmacological profile at standard concentrations may be more limited than previously assumed.
Simultaneously, the released nicotinic acid exerts vasodilatory effects on cerebral blood vessels by stimulating prostaglandin production and activating the hydroxycarboxylic acid receptor 2 (HCA2/GPR109A). This vasodilation increases cerebral blood flow, potentially enhancing nutrient and oxygen delivery to brain tissue. The dual mechanism of GABAergic inhibition combined with improved cerebral perfusion underlies picamilon's reported nootropic and anxiolytic properties.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Modest Potency:: Generally considered less effective than phenibut or prescription anxiolytics, with users describing it as providing subtle rather than dramatic anxiety relief. Individual Variation:: Responses vary significantly - some users find 100mg effective while others notice minimal benefits even at higher doses. Synergistic Effects:: Often stacked with other compounds (fasoracetam, tianeptine, L-theanine) where users report enhanced effectiveness compared to standalone use. Short Duration:: Half-life of only 1-2 hours requires multiple daily doses, making it less practical and more expensive than longer-acting alternatives
- Generally considered less effective than phenibut or prescription anxiolytics, with users describing it as providing subtle rather than dramatic anxiety relief
- Responses vary significantly - some users find 100mg effective while others notice minimal benefits even at higher doses
- Often stacked with other compounds (fasoracetam, tianeptine, L-theanine) where users report enhanced effectiveness compared to standalone use
- Half-life of only 1-2 hours requires multiple daily doses, making it less practical and more expensive than longer-acting alternatives
Safety Profile
Side effects may include headaches, dizziness, nausea, and skin flushing, particularly at higher doses. It is contraindicated in individuals with severe hypotension, renal disease, or hypersensitivity to niacin. Picamilon should not be combined with benzodiazepines or other GABAergic drugs without medical supervision, and it is not recommended during pregnancy or lactation.
Pharmacokinetic Profile
Quick Start
- Typical Dose
- Most users report taking 50-100mg, 2-3 times daily, with 100mg being the most commonly mentioned effective dose
Molecular Structure
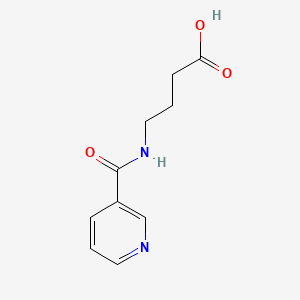
- Formula
- C10H12N2O3
- Weight
- 208.21 Da
- PubChem CID
- 60608
- Exact Mass
- 208.0848 Da
- LogP
- -0.4
- TPSA
- 79.3 Ų
- H-Bond Donors
- 2
- H-Bond Acceptors
- 4
- Rotatable Bonds
- 5
- Complexity
- 231
Identifiers (SMILES, InChI)
InChI=1S/C10H12N2O3/c13-9(14)4-2-6-12-10(15)8-3-1-5-11-7-8/h1,3,5,7H,2,4,6H2,(H,12,15)(H,13,14)
NAJVRARAUNYNDX-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Minimal Reported Issues:: Users generally report fewer side effects compared to phenibut or benzodiazepines, with good tolerability at recommended doses
- Tolerance Development:: Some users note diminishing effects with daily use, though less severe than phenibut tolerance
- Dependency Concerns:: While considered less addictive than phenibut, users caution against daily long-term use of any GABAergic compound
- Individual Reactions:: Occasional reports of headaches or ineffectiveness, though serious adverse effects are rarely mentioned in user experiences
References (6)
- [1]Identification and quantification of vinpocetine and picamilon in dietary supplements sold in the United States
→ Study found picamilon being sold illegally in US dietary supplements despite never receiving FDA approval, raising regulatory and safety concerns about unapproved drugs in the supplement market.
- [2]Picamilon, a γ-aminobutyric acid (GABA) analogue and marketed nootropic, is inactive against 50 biological targets
→ In vitro screening revealed picamilon showed weak or no binding to 50 safety-related biological targets (receptors, ion channels, enzymes) at 10 μM, suggesting a different binding profile than other GABA analogues despite structural similarity.
- [3]Gabaergic mechanism of cerebrovascular and neuroprotective effects of afobazole and picamilon
→ Research in rats found that picamilon's cerebrovascular effects were not affected by GABA receptor blocking but were significantly reduced by blocking chloride channels, suggesting its mechanism involves GABA chloride channel activity.
- [4]The new cerebrovascular preparation pikamilon
→ Early Russian research demonstrated picamilon significantly increased cerebral blood flow in conscious cats and inhibited neurogenic spasms of cerebral vessels while suppressing sympathetic nerve activity.
- [5]Clinical efficacy and safety of Picamilon in patients with progressive chronic cerebral ischemia
→ A 2024 Russian clinical study of 50 patients with chronic cerebral ischemia found picamilon improved cognitive status, reduced neurological disorders, and enhanced sleep quality with good safety profile over 70 days of treatment.
- [6]Integrative assessment of the effectiveness and safety of outpatient use of Picamilon
→ Open randomized trial of 44 patients with stage I chronic cerebral ischemia showed picamilon improved cognitive function and vegetative symptoms both with oral-only and combined parenteral/oral administration over 60 days.
Phylloquinone
Phylloquinone (Vitamin K1) is a fat-soluble vitamin essential for blood clotting and bone metabolism, functioning as a coenzyme for vitamin K-dependent carboxyl
Piceatannol
Piceatannol is a natural polyphenolic compound and hydroxylated analog of resveratrol found in various plants, particularly grapes and passion fruit. It functio