Atomoxetine
Atomoxetine is a selective norepinephrine reuptake inhibitor approved for the treatment of attention-deficit/hyperactivity disorder in both children and adults.
Overview
Atomoxetine (marketed as Strattera) is a selective norepinephrine reuptake inhibitor (NRI) and the first non-stimulant medication approved by the FDA for the treatment of attention-deficit/hyperactivity disorder (ADHD). Unlike stimulant medications such as methylphenidate or amphetamines, atomoxetine does not act on the dopamine transporter directly, instead selectively blocking the norepinephrine transporter (NET) in the prefrontal cortex. Because NET also transports dopamine in prefrontal regions, atomoxetine indirectly increases dopamine availability in this area without significantly affecting dopamine in the nucleus accumbens, which contributes to its low abuse potential.
Clinical trials have demonstrated that atomoxetine significantly reduces ADHD symptoms relative to placebo in both pediatric and adult populations, though its effect size is generally considered smaller than that of stimulant medications. The therapeutic onset is gradual, typically requiring four to six weeks for full efficacy, which distinguishes it from the rapid onset of stimulants. It is often considered when stimulants are contraindicated, poorly tolerated, or when there is concern about substance abuse risk. Off-label uses have included treatment of executive function deficits and comorbid anxiety disorders.
Common side effects include decreased appetite, nausea, dry mouth, insomnia, and dizziness. The FDA has issued a black box warning regarding an increased risk of suicidal ideation in children and adolescents. Atomoxetine is primarily metabolized by CYP2D6, and poor metabolizers of this enzyme may experience higher plasma concentrations and increased side effects. It is classified as a non-controlled substance in most jurisdictions.
Mechanism of Action
Selective Norepinephrine Reuptake Inhibition
Atomoxetine (Strattera) is a highly selective norepinephrine transporter (NET) inhibitor with a Ki of approximately 5 nM. It blocks the presynaptic NET, preventing reuptake of norepinephrine (NE) from the synaptic cleft and increasing extracellular NE concentrations. Unlike psychostimulants, atomoxetine has negligible affinity for dopamine transporter (DAT), serotonin transporter (SERT), or monoamine receptors at therapeutic concentrations (PMID: 12404710).
Prefrontal Cortex Dopamine Enhancement
In the prefrontal cortex (PFC), where DAT expression is sparse, NET is the primary mechanism for dopamine clearance. By blocking NET, atomoxetine simultaneously increases both norepinephrine and dopamine levels in the PFC — the critical region for executive function, working memory, and attention regulation. This dual catecholamine enhancement in the PFC underlies its therapeutic efficacy in ADHD without increasing dopamine in the nucleus accumbens (reward center) (PMID: 12151506).
Alpha-2 Adrenergic Receptor Signaling
Elevated NE activates postsynaptic alpha-2A adrenergic receptors on PFC pyramidal neurons, strengthening network connectivity for working memory and top-down attentional control. This mechanism is shared with guanfacine and is critical for reducing distractibility and improving sustained attention. Simultaneously, alpha-1 receptor activation at optimal levels enhances signal-to-noise ratio in PFC circuits (PMID: 21366547).
Non-Stimulant Pharmacological Profile
Unlike methylphenidate or amphetamine, atomoxetine does not significantly increase dopamine in the ventral striatum/nucleus accumbens, explaining its low abuse potential and Schedule-free classification. Microdialysis studies confirm dopamine elevation is restricted to the PFC, not mesolimbic reward pathways (PMID: 11865310).
Gradual Onset & Neuroplastic Effects
Full therapeutic effect requires 4-6 weeks, suggesting involvement of CREB-mediated gene expression changes, BDNF upregulation, and synaptic remodeling in prefrontal circuits beyond acute catecholamine modulation.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Poor Compared to Stimulants:: Consistently rated as less effective than methylphenidate, amphetamines, or even modafinil for ADHD symptoms. Individual Variability:: A small subset reports positive effects when combined with other medications (bupropion, probiotics), but most find it unhelpful. Executive Function:: Limited improvement in executive dysfunction, motivation, or working memory compared to expectations. Long-Term Tolerance:: Even when initially effective, benefits may diminish over time without dosage adjustment
- Consistently rated as less effective than methylphenidate, amphetamines, or even modafinil for ADHD symptoms
- A small subset reports positive effects when combined with other medications (bupropion, probiotics), but most find it unhelpful
- Limited improvement in executive dysfunction, motivation, or working memory compared to expectations
- Even when initially effective, benefits may diminish over time without dosage adjustment
Safety Profile
Safety Profile: Atomoxetine (Strattera)
Common Side Effects
- Decreased appetite and weight loss (10-25% of patients)
- Nausea, vomiting, and abdominal pain (reported in 12-20% of patients)
- Dry mouth (reported in ~19% of adults)
- Insomnia and sleep disturbance (15-19%)
- Fatigue and somnolence (8-11%)
- Dizziness (5-11%)
- Constipation (8% of adults)
- Urinary hesitancy and retention (6% of adults)
- Sexual dysfunction: decreased libido, erectile dysfunction, ejaculatory difficulty (reported in 2-7% of adult males)
- Increased heart rate (5-10 bpm average increase) and blood pressure (2-4 mmHg systolic)
- Mood swings, irritability
- Sweating and hot flushes
Serious Adverse Effects
- FDA Black Box Warning - Suicidal Ideation: Increased risk of suicidal thinking and behavior in children and adolescents with ADHD (0.4% atomoxetine vs 0% placebo in pooled analyses). Close monitoring required, especially during first months and dose changes
- Severe hepatotoxicity: Rare but potentially life-threatening hepatic failure reported. Discontinue immediately if jaundice or laboratory evidence of liver injury develops. Incidence estimated at 1 in 50,000-100,000 patient-years
- Serious cardiovascular events: Sudden death reported in patients with pre-existing structural cardiac abnormalities. Tachycardia, hypertension, orthostatic hypotension
- Priapism: Rare reports of prolonged painful erections; requires emergency treatment
- Severe allergic reactions including angioedema and urticaria
- Raynaud's phenomenon (new onset or worsening)
- Psychosis and mania: New-onset psychotic or manic symptoms in patients without prior history
- Seizures: Reported in <1% of patients; use with caution in patients with seizure history
- Growth retardation in children: Mean height and weight deficits of approximately 0.4 cm/year and 0.5 kg/year
Contraindications
- Known hypersensitivity to atomoxetine
- Concomitant use with MAO inhibitors or within 14 days of MAOI discontinuation (risk of hypertensive crisis)
- Narrow-angle glaucoma
- Pheochromocytoma or history of pheochromocytoma
- Severe cardiovascular or cerebrovascular disorders where blood pressure or heart rate increases would be clinically concerning
- Current or history of bipolar disorder without adequate mood stabilization
Drug Interactions
- MAO inhibitors (phenelzine, tranylcypromine, selegiline, linezolid): CONTRAINDICATED. Risk of hypertensive crisis, hyperthermia, and serotonin syndrome-like reactions
- CYP2D6 inhibitors (fluoxetine, paroxetine, quinidine, bupropion): Significantly increase atomoxetine exposure (6-8 fold in extensive metabolizers). Dose reduction required
- Pressor agents (pseudoephedrine, phenylephrine): Additive cardiovascular effects; use with caution
- Beta-2 agonists (albuterol): Potentiated cardiovascular effects (tachycardia, blood pressure elevation)
- Antihypertensives: Atomoxetine may reduce efficacy; monitor blood pressure
- Other noradrenergic agents (venlafaxine, duloxetine): Additive noradrenergic effects; monitor for cardiovascular changes
- QT-prolonging drugs: Caution advised though atomoxetine has minimal direct QT effect
Population-Specific Considerations
- Pregnancy (Category C): Decreased pup survival and weight in animal studies at doses ≥human therapeutic exposure. No adequate human studies. Use only if potential benefit justifies risk. Registry data (limited) has not identified a pattern of major birth defects. Discontinue or reassess during pregnancy planning
- Pediatric (≥6 years FDA-approved): Black Box Warning for suicidal ideation in children/adolescents. Monitor growth (height and weight) quarterly. Approved for ages ≥6 years. Not recommended under age 6 due to insufficient safety data. Weight-based dosing: start 0.5 mg/kg/day, target 1.2 mg/kg/day, max 1.4 mg/kg/day or 100 mg
- Elderly: Limited clinical trial data in patients >65. Increased risk of orthostatic hypotension and urinary retention. Start at lowest dose. Hepatic metabolism may be reduced. CYP2D6 phenotype testing may be useful for dose optimization. Cardiovascular monitoring essential
Pharmacokinetic Profile
Atomoxetine — Pharmacokinetic Curve
SubcutaneousMolecular Structure
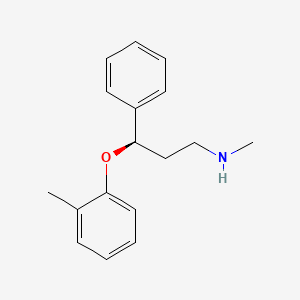
- Formula
- C17H21NO
- Weight
- 255.35 Da
- PubChem CID
- 54841
- Exact Mass
- 255.1623 Da
- LogP
- 3.7
- TPSA
- 21.3 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 2
- Rotatable Bonds
- 6
- Complexity
- 237
Identifiers (SMILES, InChI)
InChI=1S/C17H21NO/c1-14-8-6-7-11-16(14)19-17(12-13-18-2)15-9-4-3-5-10-15/h3-11,17-18H,12-13H2,1-2H3/t17-/m1/s1
VHGCDTVCOLNTBX-QGZVFWFLSA-NSafety Profile
Common Side Effects
- Personality Changes:: Emotional numbing, apathy, and feeling 'dumb' or cognitively foggy are frequently reported
- Physical Effects:: Restlessness, tension, TMJ from muscle tension, increased heart rate, and urinary retention in some users
- Sleep Disruption:: Can cause insomnia regardless of dosing time, with some reporting inability to sleep for extended periods
- Cardiovascular Concerns:: May cause QT prolongation and increased cardiovascular burden, particularly at higher doses or in sensitive individuals
References (6)
- [1]Attention-deficit-hyperactivity disorder: an update
→ Comprehensive review showing that drug therapy including atomoxetine is effective for all age groups with ADHD, though stimulants like methylphenidate and amphetamine remain the most effective first-line treatments.
- [4]Assessment of probiotic strain Lactobacillus acidophilus LB supplementation as adjunctive management of attention-deficit hyperactivity disorder in children and adolescents
→ Randomized controlled trial showing probiotics as adjunctive therapy with atomoxetine may enhance ADHD symptom management and cognitive function in children and adolescents.
- [2]An update on pharmacotherapy of autism spectrum disorder in children and adolescents
→ Evidence supports use of atomoxetine for ADHD symptoms in children and adolescents with autism spectrum disorder, though it does not address core autism symptoms.
- [3]Effects of stimulants and atomoxetine on emotional lability in adults: A systematic review and meta-analysis
→ Meta-analysis examining effects of atomoxetine on emotional lability symptoms in adults with ADHD, comparing its effects with those on core ADHD symptoms.
- [5]Metabolic Activation of Atomoxetine Mediated by Cytochrome P450 2D6
→ Study characterizing reactive metabolites of atomoxetine and potential mechanisms of hepatotoxicity, identifying glutathione and N-acetylcysteine conjugates in liver microsomes.
- [6]Amphetamine positive urine toxicology screen secondary to atomoxetine
→ Case report documenting the first instance of atomoxetine causing false-positive amphetamine results on urine drug immunoassays, though not confirmed by gas chromatography-mass spectrometry.
Astragalus Polysaccharides
Astragalus polysaccharides (APS) are bioactive compounds extracted from Astragalus membranaceus, a traditional Chinese medicinal herb. These complex carbohydrat
B7-33
B7-33 is a synthetic single-chain peptide derived from the B-chain of human relaxin-2 (H2-relaxin). It was designed to retain the anti-fibrotic properties of re