MELANOTAN 2
Melanotan 2 (MT-2) is a synthetic cyclic heptapeptide analog of alpha-MSH that acts as a non-selective melanocortin receptor agonist, studied for effects on skin pigmentation, sexual function, appetite regulation, and metabolic signaling.
Overview
MT-2 was originally developed at the University of Arizona as a tool for studying melanocortin receptor signaling. Its broad receptor activity has made it a subject of research in diverse fields ranging from dermatology and sexual medicine to neuroscience and metabolic regulation. As a cyclic peptide analog, MT-2 exhibits greater metabolic stability than its linear parent compound alpha-MSH, contributing to a longer duration of action and increased receptor binding affinity.
Mechanism of Action
MT-2 binds to melanocortin receptors (MC1R through MC5R) with varying affinities. Activation of MC1R in melanocytes stimulates melanogenesis and pigmentation. Activation of MC4R in the hypothalamus modulates appetite, energy homeostasis, and sexual arousal pathways. MC3R activation is implicated in energy balance and nutrient partitioning. The peptide's effects on sexual function are mediated primarily through central MC4R signaling, which triggers downstream pathways involving oxytocin and dopamine release.
Reconstitution Calculator
MELANOTAN 2
Melanotan 2 (MT-2) is a synthetic cyclic heptapeptide analog of alpha-melanocyte

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Systematic Truncation Studies
Hruby et al. (1995) conducted definitive SAR studies establishing the minimal pharmacophore. Sequential removal of N-terminal (Ser1-Met4) and C-terminal (Gly10-Val13) residues from alpha-MSH demonstrated that biological activity was retained in fragments containing residues 6-9 (His-Phe-Arg-Trp). Activity was abolished when any of these four residues was deleted or replaced with alanine, confirming their essential role.
D-Amino Acid Substitution and the Superpotent Agonist
The substitution of L-Phe7 with D-Phe7 was first reported by Sawyer et al. (1980), who demonstrated that [Nle4, D-Phe7]-alpha-MSH (later designated afamelanotide/Melanotan I) exhibited 10-100 fold greater potency than native alpha-MSH in frog skin melanophore bioassays. The D-Phe substitution stabilizes a type II beta-turn in the pharmacophore region, locking the peptide backbone into the bioactive conformation. This conformational constraint simultaneously enhances binding affinity and resists enzymatic degradation by chymotrypsin-like proteases.
Cyclic Lactam Analogs and MT-II
Building on the linear pharmacophore, Al-Obeidi et al. (1989)) designed Melanotan II by constraining the His-D-Phe-Arg-Trp core within a cyclic lactam framework: Ac-Nle-cyclo[Asp-His-D-Phe-Arg-Trp-Lys]-NH2. The Asp-Lys lactam bridge restricts conformational freedom, dramatically increasing potency (EC50 in the low nanomolar range) and metabolic stability. This design principle — constraining the linear pharmacophore within a cyclic scaffold — became the template for all subsequent melanocortin drug development.
Development of Bremelanotide (PT-141)
Bremelanotide arose from the observation that MT-II undergoes C-terminal hydrolysis in vivo, producing the des-amide metabolite Ac-Nle-cyclo[Asp-His-D-Phe-Arg-Trp-Lys]-OH. Hadley & Dorr (2006) recognized that this metabolite retained the His-D-Phe-Arg-Trp pharmacophore and exhibited relatively greater MC4R selectivity than the parent compound. This metabolite was developed as bremelanotide and received FDA approval in 2019 for hypoactive sexual desire disorder.
Development of Setmelanotide (Imcivree)
Kievit et al. (2013) described the design of setmelanotide, a cyclic octapeptide that incorporates the His-D-Phe-Arg-Trp core with modified flanking residues optimized for high MC4R selectivity (>20-fold over MC3R and >1000-fold over MC1R). This selectivity minimizes pigmentation side effects while maximizing the appetite-suppressing activity mediated through hypothalamic MC4R. Setmelanotide received FDA approval in 2020 for rare genetic obesity disorders.
Receptor Subtype Selectivity from Flanking Residues
While the core tetrapeptide is necessary for activity at all MCRs, selectivity between receptor subtypes is determined by flanking residues and macrocyclic constraints. Grieco et al. (2002) demonstrated through systematic analog synthesis that: MC1R selectivity is enhanced by linear extensions mimicking alpha-MSH; MC4R selectivity is achieved through specific cyclic constraints and D-amino acid substitutions flanking the core; MC3R selectivity requires distinct ring sizes and stereochemistry. This understanding enabled rational design of receptor-selective drugs from the common pharmacophore.
Neurodevelopmental Behavior Models
In a maternal immune activation (MIA) mouse model, MT-2 administration was reported alongside changes in measured autism-like behavioral endpoints and oxytocin-related signaling readouts, supporting continued investigation of melanocortin-linked circuitry in social-behavior paradigms. Studies have demonstrated that MT-2 returns sociability ratings to near baseline levels in control animals, suggesting potential relevance of melanocortin signaling to social behavior regulation (Minakova et al., 2019).
Feeding Behavior and Macronutrient Preference
Human and animal research has examined melanocortin signaling, including MC4R-linked paradigms, for relationships to dietary intake patterns and macronutrient preference (Kishi et al., 2003). Additional literature discusses leptin-melanocortin network interactions relevant to feeding and energy expenditure regulation (Cone, 2005). The melanocortin system plays a central role in integrating peripheral energy signals with central appetite regulation, making MT-2 a valuable pharmacological tool for dissecting these pathways.
Metabolic Regulation and Glucose Handling
Metabolic research has explored CNS-linked signaling contributions to peripheral glucose handling. In hypothalamic-nuclei injection models, melanocortin receptor agonism has been evaluated for effects on glucose uptake endpoints in peripheral tissues (Toda et al., 2009). These findings support ongoing interest in the central regulation of metabolic function through melanocortin pathways, with implications for understanding type 2 diabetes pathophysiology.
Reward and Impulse-Related Behavioral Research
Rodent studies have reported that melanocortin signaling in regions such as the amygdala may influence alcohol intake behaviors, with MT-2 used as a receptor-agonist probe (Navarro et al., 2011). Additional work has evaluated combined-intervention designs where MT-2 is assessed alongside naltrexone in binge-like ethanol intake models in mice (Navarro et al., 2015), demonstrating potential synergistic effects on reducing alcohol consumption.
Sexual Function
Clinical research has evaluated MT-2 in the context of psychogenic erectile dysfunction. A placebo-controlled crossover study reported significant effects on erection initiation, supporting the role of central melanocortin signaling in sexual arousal (Wessells et al., 1998). This line of research contributed directly to the development of PT-141 (bremelanotide), a more selective MC4R agonist designed specifically for sexual dysfunction.
Safety Profile
Research in humans has documented several common effects associated with MT-2 administration, including nausea, facial flushing, and fatigue following injection. Spontaneous penile erections have been frequently reported in male subjects. Hyperpigmentation, including darkening of existing nevi, has been observed and raises concern regarding dermatological monitoring. Studies have noted occasional cardiovascular effects including transient increases in blood pressure. Long-term safety data in humans remains limited, and the peptide has not received regulatory approval for clinical use. Research subjects with a history of melanoma or cardiovascular disease are typically excluded from studies due to theoretical risks.
Pharmacokinetic Profile
MELANOTAN 2 — Pharmacokinetic Curve
Subcutaneous injectionQuick Start
- Typical Dose
- 0.5-1mg per injection.
- Route
- Subcutaneous injection
- Storage
- Refrigerate 2-8°C
Molecular Structure
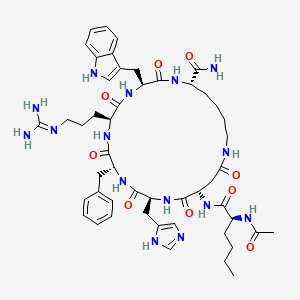
- Formula
- C50H69N15O9
- Weight
- 1024.18 Da
- CAS
- 121062-08-6
- PubChem CID
- 92432
- Exact Mass
- 1023.5403 Da
- LogP
- 0
- TPSA
- 385 Ų
- H-Bond Donors
- 13
- H-Bond Acceptors
- 11
- Rotatable Bonds
- 17
- Complexity
- 1950
Identifiers (SMILES, InChI)
InChI=1S/C50H69N15O9/c1-3-4-16-36(59-29(2)66)44(69)65-41-25-42(67)55-20-11-10-18-35(43(51)68)60-47(72)39(23-31-26-57-34-17-9-8-15-33(31)34)63-45(70)37(19-12-21-56-50(52)53)61-46(71)38(22-30-13-6-5-7-14-30)62-48(73)40(64-49(41)74)24-32-27-54-28-58-32/h5-9,13-15,17,26-28,35-41,57H,3-4,10-12,16,18-25H2,1-2H3,(H2,51,68)(H,54,58)(H,55,67)(H,59,66)(H,60,72)(H,61,71)(H,62,73)(H,63,70)(H,64,74)(H,65,69)(H4,52,53,56)/t35-,36-,37-,38+,39-,40-,41-/m0/s1
JDKLPDJLXHXHNV-MFVUMRCOSA-NResearch Indications
Dermatological
Stimulates eumelanin production via MC1R agonism, inducing skin darkening. Phase I clinical trial demonstrated tanning with 5 subcutaneous doses (PMID: 8637402). Not FDA-approved.
Hormonal
Potent initiator of penile erection in men with ED via MC4R activation. Human clinical studies showed increased rigidity and duration (PMID: 11035391, 15996790). Not approved; led to bremelanotide development.
Increased sexual desire and genital arousal in women in clinical studies. MC4R-mediated central mechanism (PMID: 15996790). Not approved; succeeded by bremelanotide.
Research Protocols
subcutaneous Injection
Protocol: 0.010-0.025 mg/kg administered subcutaneously daily for 5-14 days in fair-skinned (Fitzpatrick skin types I-II) male volunteers (n=40). Subcutaneous MT-2 at 0.025 mg/kg was administered as a single dose, with RigiScan monitoring for 6 hours post-injection.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| General Research Protocol | 0.010-0.025 mg, 0.020 mg, 0.025 mg | Daily | 2-3 weeks(Route: Subcutaneous Injection, Intracerebroventricular) |
intranasal Injection
Intranasal absorption has been explored but shows erratic bioavailability (~5-15%) similar to PT-141's intranasal pharmacokinetics.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Was observed at | 0.020 mg | Per protocol | 2-3 weeks |
| Mice | 1.0 mg | Per protocol | — |
| General Research Protocol | 0.5-1.0 mg | Daily | 5-14 days |
oral
The peptide is not orally bioavailable due to rapid gastrointestinal proteolysis.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Was observed at | 0.020 mg | Per protocol | 2-3 weeks |
| Mice | 1.0 mg | Per protocol | — |
| General Research Protocol | 0.5-1.0 mg | Daily | 5-14 days |
intracerebroventricular Injection
Subsequent protocols in rodent models used intracerebroventricular (ICV) injection of MT-2 at 0.5-5.0 nmol to assess food intake reduction over 4-24 hour periods.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| General Research Protocol | 0.010-0.025 mg, 0.020 mg, 0.025 mg | Daily | 2-3 weeks(Route: Subcutaneous Injection, Intracerebroventricular) |
Interactions
Peptide Interactions
Navarro et al. (2015) demonstrated synergistic effects of MT-2 (0.5 nmol ICV) and naltrexone (1.0 mg/kg IP) on reducing binge-like ethanol intake in mice. The combination produced greater reduction (~70%) than either agent alone (MT-2: ~30%, naltrexone: ~40%), suggesting melanocortin and opioid s...
The peptide's effects on sexual function are mediated primarily through central MC4R signaling, which triggers downstream pathways involving oxytocin and dopamine release.
MT-II Fragment is derived from Melanotan II (Ac-Nle-cyclo[Asp-His-D-Phe-Arg-Trp-Lys]-NH2) and retains partial melanocortin receptor activity. Co-administration with full-length MT-II would be redundant and could potentially compete for MC receptor binding. Use of both simultaneously is unnecessary. (Hadley, 2005, Peptides)
What to Expect
What to Expect
Effects begin within hours of administration based on half-life of ~1-2 hours
MT-2 produced clinically meaningful erections (>60% rigidity at tip and base lasting >10 minutes) in 8 of 10 subjects vs.
Significant darkening of skin was observed at 0.020 mg/kg and above within 5 days, with effects persisting 2-3 weeks after cessation.
Protocol: 0.010-0.025 mg/kg administered subcutaneously daily for 5-14 days in fair-skinned (Fitzpatrick skin types I-II) male volunteers (n=40).
Effects persist for 2-4 weeks after discontinuation because eumelanin deposited in keratinocytes is slowly lost through normal epidermal turnover...
Safety Profile
Common Side Effects
- Nausea:: Very common, especially initially.
- Flushing:: Facial flushing after injection.
- Mole Changes:: Darkening and potential changes to moles.
- Spontaneous Erections:: Can be inconvenient.
- Blood Pressure:: May affect cardiovascular system.
Quality Indicators
What to look for
- Extensive peer-reviewed research base
- Oral administration available
Caution
- Short half-life may require frequent dosing
Frequently Asked Questions
References (14)
- [13]Patel et al — Understanding melanocortin receptors: a comprehensive review of their biology and therapeutic potential Peptides (2023)
- [2]Melanotan-II reverses memory impairment induced by a short-term HF diet
→ Demonstrates that Melanotan-II reversed negative neurobehavioral effects caused by high-fat diet in zebrafish models.
- [3]Melanocortin receptor agonist melanotan-II microinjected in the nucleus accumbens decreases appetitive and consumptive responding for food
→ Found that melanotan-II injection reduced food intake motivation in mice without causing adverse effects.
- [4]The potent melanocortin receptor agonist melanotan-II promotes peripheral nerve regeneration and has neuroprotective properties in the rat
→ Tested melanotan-II for nerve recovery in rats, finding significant sensory function improvement and cisplatin toxicity protection.
- [11]
- [6]Wessells et al *J Urol* J Urol (1998)
- [7]Navarro et al *Br J Pharmacol* Br J Pharmacol (2011)
- [9]
- [10]
- [5]The protective effects of the melanocortin receptor agonist, melanotan-II, against binge-like ethanol drinking are facilitated by deletion of the MC3 receptor in mice
→ Found melanotan-II successfully reduces binge alcohol consumption through specific receptor mechanisms.
- [8]
- [12]Ericson et al — Melanocortin 4 receptor-mediated anorexia involves multiple signaling pathways J Endocrinol (2022)
- [14]Rodrigues et al — Melanocortin system dysregulation in obesity and metabolic diseases Eur J Pharmacol (2022)
- [1]Melanotan II: a possible cause of renal infarction: review of the literature and case report
→ Examines renal infarction as a rare complication potentially induced by Melanotan II, reviewing mechanisms including blood clot formation.
MELANOTAN 1
Melanotan I, known by its generic name afamelanotide and brand name Scenesse, is a synthetic tridecapeptide analog of alpha-melanocyte stimulating hormone (alph
Melanotan I
Synthetic analog of α-MSH that selectively targets MC1 receptors for melanin stimulation. FDA-approved as SCENESSE implant for erythropoietic protoporphyria (EP